The pV-diagram in Fig. P20.51 shows the cycle for a refrigerator operating on 0.850 mol of H
Question:
Figure P20.51
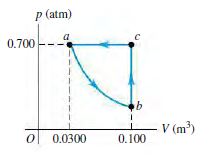
Transcribed Image Text:
p (atm) 0.700 V (m) ol 0.0300 0.100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
IDENTIFY and SET UP A refrigerator is like a heat engine run in reverse In the pVdiagram shown with ...View the full answer

Answered By

Talha Talib
I am a member of IEEE society. As i am a student of electrical engineering badge 17 but beside of this i am also a tutor in unique academy. I teach calculus, communication skills, mechanics and economics. I am also a home tutor. My student Muhammad Salman Alvi is a brilliant A-level student and he performs very well in academics when i start to teach him. His weak point was mathematics but now he is performing well in mathematics. I am a scholarship holder in Fsc as i scored 1017 marks in metric out of 1100. Later on i got scholarship in Punjab Group of Colleges. I got 2nd position in robotics competition in 2018 as my project home automation select for the exhibition in Expocentre.
4.60+
23+ Reviews
62+ Question Solved
Related Book For 

University Physics with Modern Physics
ISBN: 978-0133977981
14th edition
Authors: Hugh D. Young, Roger A. Freedman
Question Posted:
Students also viewed these Physics questions
-
The PV diagram in Fig. 15-23 shows two possible states of a system containing 1.35 moles of a monatomic ideal gas, (P1 = P2 = 455 N/m2, V1 = 2.00 m3, V2 = 8.00m3.) (a) Draw the process which depicts...
-
The PV diagram in Fig. 15-23 shows two possible states of a system containing 1.75 moles of a monatomic ideal gas. (P1 = P2 = 425 N / m2, V1 = 2.00 m3, V2 = 8.00 m3.) (a) Draw the process which...
-
Two moles of a diatomic gas are taken through the cycle ABCA as shown on the PV diagram in figure. At A the pressure and temperature are 5 atm and 600 K. The volume at B is twice that at A. The...
-
Simplify each expression in Problems 722. Classify each answer by number of terms and degree. 3(x5) 2(x + 8)
-
Two charges are placed as shown in Fig. 17-49 with q1 = 1.2 μC and q2 = - 3.3 μC. Find the potential difference between points A and B. A 0.10 m 92 -0.10 m-
-
The Fisher effect says that nominal interest rates will equal expected inflation plus the real equilibrium rate of return: i = EÏ + rEquilibrium (2) i = Nominal interest rate, EÏ = Expected...
-
What are the main factors considered when writing a report?
-
One of the more closely watched ratios by investors is the price/earnings or P/E ratio. By dividing price per share by earnings per share, analysts get insight into the value the market attaches to a...
-
If today's 1-year interest rate is 5%, and you expect 1-year interest rates to be 6% next year and 6.25% the year after that, compute the term structure and draw today's yield curve based on the...
-
Show that (n+1) 5 is O(n 5 ).
-
Premium gasoline produces 1.23 10 8 J of heat per gallon when it is burned at approximately 400C (although the amount can vary with the fuel mixture). If a cars engine is 25% efficient,...
-
In your summer job with a venture capital firm, you are given funding requests from four inventors of heat engines. The inventors claim the following data for their operating prototypes: (a) Based on...
-
Solve the given equation using an integrating factor. Take t > 0. y = .5(35 - y)
-
Is the drug court method of dealing with nonviolent drug offenders superior to the traditional methods of imposing a fine or jail term or placing the defendant on probation? Should drug courts limit...
-
In New Jersey v. T.L.O. (1985), the Supreme Court adopted a reasonableness standard for public school searches. Should this standard be applied to searches of students in public colleges and...
-
What justifies the criminal law making mere possession of contraband articles illegal? Do you think this rationale extends to criminalizing the mere possession of such innocent items as a...
-
Why, according to Robinson v. California, may a person not be held criminally liable for being addicted to illicit narcotics? Is this decision distinguishable from Powell v. Texas?
-
Today many security personnel are private police, yet Fourth Amendment protection has been extended only to those searches conducted by government officials. What arguments can be made for and...
-
Which mortgage would result in higher total payments? Mortgage A: $985 a month for 30 years Mortgage B: $780 a month for 5 years and $1,056 for 25 years
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
An echo is sound reflected from a distant object, such as a wall or a cliff. Explain how you can determine how far away the object is by timing the echo?
-
Why do you see lightning before you hear the thunder? A familiar rule of thumb is to start counting slowly, once per second, when you see the lightning; when you hear the thunder, divide the number...
-
Children make toy telephones by sticking each end of a long string through a hole in the bottom of a paper cup and knot-ting it so it will not pull out. When the spring is pulled taut, sound can be...
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


