Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R +
Question:
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism.
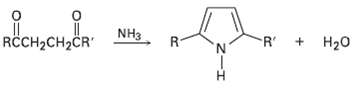
Transcribed Image Text:
R + H20 RCCH2CH2ČR" R- NH3 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
HCCH 1 RC CR 11 O HCCH 11 1 RC HA HA 0NH proton N atio...View the full answer

Answered By

Sadoc Raju
I have been teaching High School Physics in India for the last nine years of my life.
I have taught in State Board, CBSE, ICSE and IGCSE schools.
I have gained incredible experience and valuable insight over these years having worked in five different schools in two states with a diverse school population.
I have been certified by Pune University, considered to be the premier institute for education in India.
I also have worked as an aircraft technician to gain practical knowledge in mechanics and aerospace.
Why do I teach?
For me, the rewards and challenges of teaching offer a sense of satisfaction that is enhanced by the knowledge that I have had some impact on the academic and personal development of the children I teach. I believe in developing with the children a supportive classroom environment, which has a basis in quickly developing rapport with the children. I pride myself to be a dedicated self-motivated achiever who is committed to success and adapt at juggling multiple tasks in a high-pressured environment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Ammonia is a principal nitrogen fertilizer. It is prepared by the reaction between hydrogen and nitrogen. In a particular reaction, 6.0 moles of NH3 were produced. How many moles of H2 and how many...
-
Ammonia is compressed from 120 kPa with x = 1 to a pressure of 1.2 MPa and a temperature of 100C. For a mass flux of 3 kg/s, determine the power required to drive the adiabatic compressor.
-
A piston/cylinder contains 1 kg of ammonia at 20C with a volume of 0.1 m3, shown in Fig. P5.129. Initially the piston rests on some stops with the top surface open to the atmosphere, Po, so a...
-
A fast-food chain randomly attaches coupons for prizes to the packages used to serve french fries. Most of the coupons say Play again, but a few are winners. Seventy-five percent of the coupons pay...
-
In what ways does Costco meet the criteria for a "sustainable" organization?
-
Find the maximum temperature to which the following two magnesia-alumina refractory materials may be heated before a liquid phase will appear. (a) A spinel-bonded alumina material of composition 95...
-
Briefly describe the key features of a Control Risk Self Assessment (CRSA) system.
-
The Retread Tire Company recaps tires. The fixed annual cost of the recapping operation is $60,000. The variable cost of recapping a tire is $9. The company charges $25 to recap a tire. a. For an...
-
Steve's Scooters plans to sell a standard scooter for $320 and a chrome scooter for $400. Steve's purchases the standard scooter for $80 and the chrome scooter for $100. Steve's expects to sell one...
-
Review the calculations for The Diamond Ridge Golf Course example discussed in the chapter. 1. Based on these calculations, is there any good-will value? If so, how much? 2. How did you make this...
-
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount. :0: H2SO4 NH2 NH2 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
An experiment was conducted to examine the effect of age on heart rate when a person is subjected to a specific amount of exercise. Ten male subjects were randomly selected from four age groups:...
-
Dr. Sulaiman Alhabib Medical Services Group stock sells for SAR 75 per share, and you've decided to purchase 200 shares. You have $15,000 available to invest. a)Construct your equity account balance...
-
ToyWorks Ltd. is a company that manufactures and sells one single product. For planning and control purposes they utilize a monthly master budget. Their fiscal year end is December 31. It is October...
-
A firm is weighing three capacity alternatives: small, medium, and large job shop. Whatever capacity choice is made, the market for the firm's product can be "moderate" or "strong." The probability...
-
You are interested in purchasing a new Honda sedan that costs $45,000. The dealership offers you a special financing rate of 5.0% per month for 60 months. Assuming that you use your savings to make a...
-
What is the minimum number of test cases needed to meet statement coverage criterion for the following code? int total; public int show(int val) { total = 0; if (val <0)( } val = -val; while (val >...
-
You are offered $500 now plus $500 one year from now. You can earn 6 percent per year on your money. a. It is suggested that a single fair amount be paid now. What do you consider fair? b. It is...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
Write the formula for the conjugate base of each acid. a. HCI b. HSO3 c. HCHO, d. HF
-
What are all of the structural possibilities for C4H6? (Nine compounds, four acyclic and five cyclic, are known.)
-
Write an equation for each of the following reactions: a. 2-butene + HCl b. 3-hexene + HI c. 4-methylcyclopentene + HBr
-
Use Markovnikov's Rule to predict which regioisomer predominates in each of the following reactions: a. 1-pentene + HBr b. 2-methyl-2-hexene + H2O (H+ catalyst)
-
On January 1, 2023, Bertrand, Incorporated, paid $70,800 for a 40 percent interest in Chestnut Corporation's common stock. This Investee had assets with a book value of $235,000 and liabilities of...
-
In an airline's hiring process, all candidates were assessed on criteria such as "assertiveness," "teamwork" and "ability to have fun." Although these were age-neutral on their face, bias was...
-
Selected cost data for Aerial Co for the year are as follows: Estimated overhead cost for the year Estimated direct labor cost for the year Actual manufacturing overhead cost for the year Actual...

Study smarter with the SolutionInn App


