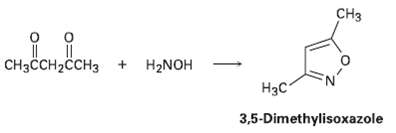
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH
Question:
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism.
Transcribed Image Text:
CHз CH3CCH2CCH3 + H2NOH Нас 3,5-Dimethylisoxazole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
This mechanism is virtually identical to the mechanism il...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following alkyl bromide can be prepared by reaction of the alcohol (S)-2-pentanol with PBr3. Name the compound, assign (R) or (S) stereochemistry, and tell whether the reaction of the alcohol...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
Mary, who is single, had the following items for 2016: Required: Determine Marys adjusted gross income for 2016. Salary $80,000 Loss on sale of 8 1244 small business stock acquired 3 years ago...
-
When a restaurant employee slipped on spilled soup and fell, requiring the evening off to recover, the owner realized that workplace safety was an issue to which she had not devoted much time. A...
-
Upon consideration of the SiO2-Al2O3 phase diagram, Figure 12.27, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices....
-
With reference to Exercise 11.16, find a \(90 \%\) confidence interval for \(\alpha\). Data From Exercise 11.16 11.16 Recycling concrete aggregate is an important compo- nent of green engineering....
-
North Pole Fishing Equipment Corporation and South Pole Fishing Equipment Corporation would have identical equity betas of 1.10 if both were all equity financed. The market value information for each...
-
What is a Living Will? What is a Health Care Proxy? What is a Durable Power of Attorney? What is an Agent for Health Care?
-
Preparation of a tax return for a partnership taxpayer Your client Nicole and Sam attended your office to have their 2 0 2 3 - year tax return prepared. Nicole Mania & Sam Baker are equal partners of...
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
Account for the fact that p-nitro aniline (pKa = 1.0) is less basic than m-nitro aniline (pKa = 2.5) by a factor of 30. Draw resonance structures to support your argument. (The PKa values refer to...
-
The sex attractant of the female arctiid moth contains, among other components, a compound of molecular formula C21H40 that yields on ozonolysis. What is the constitution of this material?...
-
The following two tables give information on treasury prices and the holdings in a portfolio managed by Bob Smith Term Structure Maturity Type Par Coupon Rate Treasury Price B spread on Treasury...
-
Printing Plus, Lynn Sanders' printing service company. We will analyze and record each of the transactions for her business and discuss how this impacts the financial statements. More detail for each...
-
46 84.8 K/s 8 (29/244) EB... token: One of the basic elements of the syntactic structure of a program, analogous to a word in a natural language. syntax: The rules that govern the structure of a...
-
Using the data below, your task is to negotiate the purchase price of the Target property with another team. All comps are within a 2 mile radius of the Target Property and represent transactions...
-
1. Signal Generation a. In PSpice, pick LM324 or uA741 (for U1 ~ U3 op-amps), construct the circuit in Figure 1. Use dual power supply of V= 9 V for the op-amps, Rref 1 and Rref_2. b. Start with the...
-
Pooi Phan needs \($2\),000 to pay off her bills. She borrows this amount from a bank with plans to pay it back over the next 4 years at $X per year. Draw a cash flow diagram from the banks...
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
In each reaction, identify the BrnstedLowry acid, the Brnsted Lowry base, the conjugate acid, and the conjugate base. H30(aq) + (aq) a. HI(aq) + HO(1) b. CH3NH(aq) + HO(1) CH3NH3 (aq) + OH (aq) c....
-
What two products are possible from the addition of HCl to 2-octene? Would you expect the reaction to be regiospecific?
-
Classify each of the following carbocations as primary, secondary, or tertiary: a. CH,CH CHCH b. (CH,) CHCH2 c. CH3
-
Write the steps in the electrophilic additions in eqs. 3.15 and 3.16, and in each case, show that reaction occurs via the more stable carbocation. CH,C CH2 H-OHCH,CCH (CH,CHCH2OH (3.15) CH3 not...
-
In the context of solid-liquid extraction, how do particle size distribution, porosity, and surface area of the solid matrix impact extraction kinetics and overall efficiency? Provide examples of...
-
Discuss the challenges and opportunities associated with the extraction of bioactive compounds from natural sources using environmentally benign solvents and green extraction techniques, such as...
-
The driver of a car traveling at 31.9 m/s applies the brakes and undergoes a constant deceleration of 1.12 m/s 2 . How many revolutions does each tire make before the car comes to a stop, assuming...

Study smarter with the SolutionInn App


