Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it
Question:
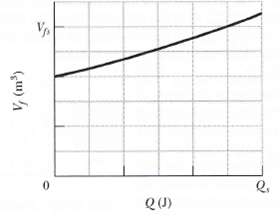
Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it as heat Q. It Figure 19-20 shows the final volume V1versus Q, what is the gas temperature? The scale of the vertical axis is set by Vfs = 0.30 m3, and the scale of the horizontal axis is set by Qs =1200J.
Transcribed Image Text:
Q()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Using Eq 1914 we note that since it is an isothermal process invol...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A quantity of an ideal gas undergoes an isothermal expansion at 20 oC and does 3.0 x 103 J of work on its surroundings in the process. (a) Will the entropy of the gas (1) increase, (2) remain the...
-
Suppose 4.00 mol of an ideal gas undergoes a reversible isothermal expansion from volume V1 to volume V2 = 2.00 V1 at temperature T = 400 K. Find (a) The work done by the gas and (b) The entropy...
-
The temperature of 0.150 mol of an ideal gas is held constant at 77.0oC while its volume is reduced to 25.0% of its initial volume. The initial pressure of the gas is 1.25 atm. (a) Determine the work...
-
The following MINITAB output exhibits the cumulative distribution function curves of three lognormal distributions. Their mean and variance, respectively, are (1, .5), (1, 1), and (1, 2). Compare the...
-
In 2014, John opened an investment account with Randy Hansen, who held himself out to the public as an investment adviser and securities broker. John contributed $200,000 to the account in 2014. John...
-
Memories Company commenced operations on July 1. Memories Company uses a periodic inventory system. During July, Memories Company was involved in the following transactions and events: July 2...
-
Comment on the statement normal flow is the best-case scenario.
-
Tess is the development manager for the Isabelle Stewart Gardner Museum in Boston. She was in the middle of a large campaign to raise $50 million for a building expansion project. Her development...
-
PATA PLC acquired 7 5 % of the equity interest in POTEA PLC at 1 January 2 0 1 9 for KShs . 5 0 million. The Statement of Financial Position as at 3 1 March 2 0 2 0 were as follows: PATA PLC POTEA...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
Water bottle in a hot car in the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a refrigerator...
-
In the temperature range 310 K to 330 K, the pressure p of a certain non ideal gas is related to volume V and temperature T by how much work is done by the gas if its temperature is raised from 315 K...
-
Determine the missing amounts
-
Use the simplex method to find the maximum value of P = 2x + y subject to these constraints. In order to correctly answer some of the following questions, you will need to use the method taught in...
-
1. not? a) State the domain and range of the following graph. Does it represent a function? Why or why 2. Let f(t)=-4t + 3t-8 b) I
-
3. For all the parts below, suppose that f : A B and let D be a subset of B. (a) Prove or give a counterexample: [(D)] D. (b) Prove or give a counterexample: D [(D)]. (c) State which of the...
-
The region W is the cone shown below. The angle at the vertex is w/3, and the top is flat and at a height of 2/3. Write the limits of integration for fw dV in the following coordinates (do not reduce...
-
Lorilari Corp, is a logistics and transportation company. Below is a table containing data stored by the company. Vehicle registration cle number type MEN3456 Van AK01 MEN3456 Van AK01 BCC9320 Vehi...
-
(a) Find all elements of the moment of inertia matrix for a cube of mass \(M\) and edge length \(\ell\) using its principal axes. (b) Then find all elements of the moment of inertia matrix for the...
-
Question 2 For an n x n matrix A = form) via (aij)
-
If you deposit $2,000 in a 5-year certificate of deposit at 5.2%, how much will it be worth in five years?
-
At the surface of Jupiter's moon 10, the acceleration due to gravity is g = 1.81 mfs2. A watermelon weighs 44.0 N at the surface of the earth. (a) What is the watermelon's mass on the earth's...
-
An astronaut's pack weighs 17.5 N when she is on earth but only 3.24 N when she is at the surface of an asteroid. (a) What is the acceleration due to gravity on this asteroid? (b) What is the mass of...
-
World-class sprinters can accelerate out of the starting blocks with an acceleration that is nearly horizontal and has magnitude 15 m/s2. How much horizontal force must a 55-kg sprinter exert on the...
-
Who is your favorite designer from the 20th Century (the 1900s)? Give a detailed explanation of why he or she is your favorite and how his/her designs have impacted the fashion industry.
-
Describe what the article was about? What stood out the most in the article? How it relates to the fashion industry? Was the article useful in expanding knowledge as it relate to textile? Website...
-
Explain why special events have arisen in human civilization, why they are so popular, and how they contribute to community development and benefit stakeholders and host communities. Describe the...

Study smarter with the SolutionInn App


