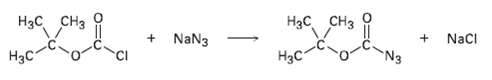
Tert-Butoxycarbonyl azide, a reagent used in protein synthesis, is prepared by treating tert-Butoxycarbonyl chloride with sodium azide.
Question:
Tert-Butoxycarbonyl azide, a reagent used in protein synthesis, is prepared by treating tert-Butoxycarbonyl chloride with sodium azide. Propose a mechanism for thisreaction.
Transcribed Image Text:
Нас, снз H3C CH3 + NaN3 NacI Нас N3 H3C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
CH33CO 0 3 addition of azide CH33CO N3 C...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide at 350 C to give a mixture of p-cresol and m-cresol.
-
Propose a mechanism for the synthesis of methyl orange.
-
A stiff, straight, horizontal wire of length 25 cm and mass 20 g is supported by electrical contacts at its ends, but is otherwise free to move vertically upward. The wire is in a uniform, horizontal...
-
Describe the advantages of face-to-face communication as opposed to interactions facilitated by technology such as telephones, e-mail, instant messaging, texting, the Web, social networking sites,...
-
George Parker was paid a salary of $74,700 during 20-- by Umberger Company. In addition, during the year, Parker started his own business as a public accountant and reported a net business income of...
-
What are the criteria used for estimating the total effort required by a salesperson to cover a territory?
-
All trucks traveling on Interstate 40 between Albuquerque and Amarillo are required to stop at a weigh station. Trucks arrive at the weigh station at a rate of 200 per 8-hour day, and the station can...
-
what is time complexity of the following code? void function ( ) { int sum = 0 ; for ( int i = 1 ; i < = 1 0 0 ; i + + ) { int x = i; while ( x ! = 0 ) { sum + = x; x / = 2 ; } } printf ( " % d " ,...
-
A circuit youre using discharges a 20 F capacitor through an unknown resistor. After charging the capacitor, you close a switch at t = 0 s and then monitor the resistor current with an ammeter. Your...
-
When ethyl benzoate is heated in methanol containing a small amount of HC1, methyl benzoate is formed. Propose a mechanism for the reaction.
-
We said in Section 21.6 that mechanistic studies on ester hydrolysis have been carried out using ethyl propanoate labeled with 180 in the ether- like oxygen. Assume that 180-labcled acetic acid is...
-
Use properties of the trigonometric functions to find the exact value of each expression. Do not use a calculator. cos 400 sec 40
-
How expense reimbursement is non-taxable to the employee? Discuss how it is added straight to the net pay when supported by receipts?
-
Risk management is one of the keys to successfully investing money because investments are never risk-free. True False
-
What happens if you change production to match sales? Change production quantities to 200,000 for each month? Can you explain how and why income comparison between absorption and variable costing has...
-
Differentiate between insurance companies and pharmaceutical companies? Explain briefly with examples.
-
Why must the cost of debt be adjusted for taxes? Multiple choice question. All sources of external financing, including the cost of debt, are adjusted for taxes Because interest on debt is tax...
-
Suppose that you have ten observations that have a mean of 7, a median of 6, and a standard deviation of 3. If you subtract 2 from the value of each observation, what are the new values of the a....
-
In Problems, solve each system of equations. x + 2y + 3z = 5 y + 11z = 21 5y + 9z = 13
-
Determine the molecular geometry and sketch each molecule or ion using the bond conventions shown in Representing Molecular Geometries on Paper in Section 11.4. a. SF 4 b. ClF 3 c. IF 2 d. IBr 4
-
Complete the reactions given in Fig. P14.45 using knowledge or intuition developed from this or previous chapters. (a) (b) CH CH MgBr DO CH,CH CH CH2 CH 0 CH CH2-O S-O- CH2CH3 diethvl sulfate
-
Complete the reactions given in Fig. P14.45 using knowledge or intuition developed from this or previous chapters. (a) (b) CH CH MgBr DO CH,CH CH CH2 CH 0 CH CH2-O S-O- CH2CH3 diethvl sulfate
-
A student consulted a well-known compilation of reference spectra for the proton NMR spectrum of propyne and was surprised to find that this spectrum consists of a single unsplit resonance at 51.8....
-
Consider a process executing on an operating system that uses demand paging. The average time for a memory access in the system is M units if the corresponding memory page is available in memory and...
-
Solve for x. log x log(x+8)=-1
-
Projectile motion is observed during the parabolic flight of an object that is thrown with an initial speed composed of upward and sideway components of velocity. The parabolic fight path can be...

Study smarter with the SolutionInn App


