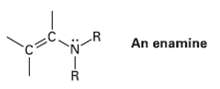
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double
Question:
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2-hybridized, draw an orbital picture of an enamine, and explain why the double bond iselectron-rich.
Transcribed Image Text:
An enamine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
The lone pair electrons from nitrogen can overlap with the double bond ele...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A routine addition of HBr across the double bond of a vinylcyclopentane gave a small amount of an unexpected rearranged product. Propose a mechanism for the formation of this product, and explain why...
-
(a) When HBr adds across the double bond of 1,2-dimethylcyclopentene, the product is a mixture of the cis and trans isomers. Show why this addition is not stereospecific. (b) When...
-
Sulfuric acid (H2SO4) adds to the double bond of alkenes as H+ and -OSO3H. Predict the products when sulfuric acid reacts with (a) Ethylene and (b) Propene.
-
Multiple Choice Questions 1. If a company has a $25,000 reduction in sales and an increase of $7,000 in fixed costs with a contribution margin ratio of 34 percent, by how much will net income change?...
-
How would you evaluate the company's method of communicating the change in pay level? What improvements to that process can you suggest?
-
Predict the type of radioactive decay process for the following radionuclides: (c) Phosphorus-32, (d) chlorine-39. B,
-
Under what conditions might an entity not responsible for dumping toxic waste be held responsible by the U.S. Environmental Protection Agency?
-
Mary Pierce is the controller of Arnold Corporation and is responsible for the preparation of the year-end financial statements. The following transactions occurred during the year. (a) On December...
-
At the bottom of a bottle a bubble within carbonated water has a diameter of 0.2 mm Determine the bubble's diameter when it reaches the surface. The temperature of the water and bubbles is 10C, and...
-
In early 20X1, Mr. Carpet purchased $200,000 of common shares in a company listed on thelocal stock exchange, with an annual eligible dividend of $5,000. The current fair market value of the shares...
-
How would you carry out the following synthesis (more than one step is required)? What stereo chemical relationship between the ?CO 2 CH 3 group attached to the cyclohexane ring and the ?CHO groups...
-
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm. How do you account for this difference? -NH2 p-Toluidine U max = 235 nm) Benzene Amax = 204 nm)
-
What's the current yield of a 5.2 percent coupon corporate bond quoted at a price of 96.78?
-
Andy currently has a balance of $4,585.92 in an account he has held for 21 years. He opened the account with an initial deposit of $3,278. What is the simple interest rate on the account?
-
The current price of DISH Network stock is $31.50 per share, and six-month European call options on the stock with a strike price of $32.50 are currently trading at $3.60. An investor, who has...
-
You short sale a common stock on margin at $40 per share with $90,000 of your own money. Assume the initial margin is 75% and the stock pays no dividend. What would the maintenance margin be if a...
-
Wilderness World (WW) plans to raise $89 million needed to pay bills by issuing new debt. To issue the debt, WW must pay its investment banker a fee equal to 2 percent of the total issue. The company...
-
A European call has strike $14 and underlying asset described by CRR notation S = 16, u = 1.02, d=0.97. For a ten-step model, what is the expiry value of this call at node (10,7)?
-
One design challenge for a capacitor-powered electric car is that the voltage would change with time as the capacitors discharged. If the capacitors in a car were discharged to half their initial...
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
A 500.0-mL sample of pure water is allowed to come to equilibrium with pure oxygen gas at a pressure of 755 mmHg. What mass of oxygen gas dissolves in the water? (The Henrys law constant for oxygen...
-
Predict the products from the reactions of the following amines with sodium nitrite in dilute HCl. (a) Cyclohexanamine (b) N-ethylhexan-2-amine (c) Piperidine (d) Aniline
-
Propose a mechanism for the synthesis of methyl orange.
-
Show how you would convert aniline to the following compounds. (a) Fluorobenzene (b) Chlorobenzene (c) 1, 3, 5-trimethylbenzene (d) Bromobenzene (e) Iodobenzene (f) Benzonitrile (g) Phenol (h) OH...
-
How has technology brought about social change in your lives in comparison to your parents' and your grandparents' generation? how has technology brought about change in your grandparents'/parents'...
-
Identify the role and function of the major components of the Canadian criminal justice system Demonstrate the manner in which the criminal justice system components relate and interact with each...
-
Design a Turing Machine (TM) for the following language, give formal description of the TM and draw state diagram: {w|w is a string over the alphabet {0, 1} and number of 1's in w is divisible by 3}....

Study smarter with the SolutionInn App


