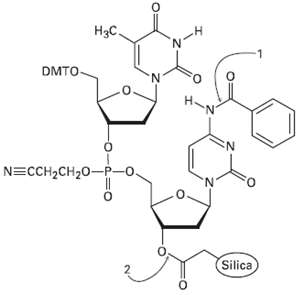
The final step in DNA synthesis is deprotection by treatment with aqueous ammonia. Show the mechanisms by
Question:
The final step in DNA synthesis is deprotection by treatment with aqueous ammonia. Show the mechanisms by which deprotection occur at the points indicated in the followingstructure:
Transcribed Image Text:
Нас. DMTO N. 'N' N=CCH2CH20-P-o. Silica
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
Both of these cleavages occur by the nowfamiliar nucleop...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The final step in the research process typically involves a memorandum to the client file and/ or a letter to the client communicating the results of the research. List the items that should be found...
-
The final step in the controlling process involves taking corrective actions if necessary. a. What types of corrective actions would you take to help control the performance of financial planners? b....
-
The orbital diagram that follows presents the final step in the formation of hybrid orbitals by a silicon atom. What type of hybrid orbital is produced in this hybridization? 1 111 1111 3s
-
In designing an experiment, the researcher can often choose many different levels of the various factors in order to try to find the best combination at which to operate. As an illustration, suppose...
-
What are the attributes associated with a successful acquisition strategy? Discuss.
-
Consider one of the cylindrical channels of inner diameter d that run through an isomerization catalyst, as shown in the figure below. A catalyst coats the inner walls of each channel. This catalyst...
-
An article in the Journal of Pharmaceutical Sciences \((\mathbf{8 0}, 971-977,1991)\) presents data on the observed mole fraction solubility of a solute at a constant temperature, along with...
-
Ashton Fleming has decided to document and analyze the accounts payable process at S&S so the transition to a computerized system will be easier. He also hopes to improve any weaknesses he discovers...
-
Over 5 periods, the arithmetic average return of a portfolio is 1.4%, the geometric average return is 1.012%, and the dollar-weighted average return is 0.577%. What might explain why the...
-
If the coefficient of static friction between all the surfaces of contact is s, determine the force P that must be applied to the wedge in order to lift the brace that supports the load F. N' PNA B....
-
Show the steps involved in a laboratory synthesis of the DNA fragment with the sequence CTAG.
-
Draw the structure of cyclic adenosine mono phosphate (cAMP), a messenger involved in the regulation of glucose production in the body. Cyclic AMP has a phosphate ring connecting the 3' and 5'...
-
Predict what will happen to stock prices after a monetary easing. Explain your prediction.
-
How might we evaluate the risk of a security when making an investment decision?
-
What would the resulting Sharpe Ratio for a portfolio formed, according to the Treynor-Black Model, by combining the market portfolio (Sharpe Ratio of 0.52) with a security that has alpha of 1.40%...
-
isabelle sterling, the partner you work for, unexpectedly had to travel to Hong Kong. She left you the Tang Aluminum Products file for your client, Global Bank. Global will be lending Tang 1.9...
-
Jimmy invests $16,000 in an account that pays 5.90% compounded quarterly. How long (in years and months) will it take for his investment to reach $21,000? ___years and ___months
-
malia bought 4 bonds of Reebox Company (11 3/4 28) at 88.25. Assuming a commission of $4 per bond, how much interest will malia recieve from the bonds after one year
-
List four types of corporate debt that are typically issued.
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
Redo Problem 6.7 using Aspen Plus Problem 6.7 One hundred cubic meters of carbon dioxide initially at 150C and 50 bar is to be isothermally compressed in a frictionless piston-and-cylinder device to...
-
Mutations (caused by radiation, cancer-producing agents, or other means) may replace one base with another or may add or delete a base. What would happen to the protein produced if the sequence UUU...
-
How many valence electrons does carbon have?
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
Solve the triangle shown to the right. Round the lengths of sides to the nearest tenth and angles to the nearest degree. 96 b=5 a=7 B A C C9.0 (Do not round until the final answer. Then round to the...
-
Hello, need help with making the program MealData.txt Avocado Egg Rolls 6.00 Parmesan-crusted Chicken 19.75 Strawberry Shortcake Pizookie 8.25 BBQ Tri-Tip Sliders 12.95 Spicy Peanut Chicken with Soba...
-
Assume you have been appointed to develop ethnic and multicultural marketing for a small chain of household appliance stores in large metropolitan area. There are several large concentrations of...
Leadership From The Inside Out Becoming A Leader For Life 3rd Edition - ISBN: 1523094354 - Free Book

Study smarter with the SolutionInn App


