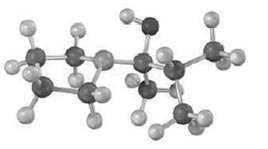
Question: The following molecular model represents a tetrahedral intermediate resulting from addition of a nucleophile to an aldehyde or ketone. Identify the reactants, and write the
The following molecular model represents a tetrahedral intermediate resulting from addition of a nucleophile to an aldehyde or ketone. Identify the reactants, and write the structure of the final product when the nucleophilic addition reaction is complete.
Step by Step Solution
3.41 Rating (164 Votes )
There are 3 Steps involved in it
The intermediate results from the addition of ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-AN (74).docx
120 KBs Word File


