The sex attractant given off by the common housefly is an alkene named muscalure. Propose a synthesis
Question:
The sex attractant given off by the common housefly is an alkene named muscalure. Propose a synthesis of muscalure starting from acetylene and any alkyl halides needed. What is the IUPAC name formuscalure?
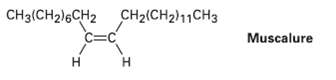
Transcribed Image Text:
CH3(CH2)6CH2 CH2(CH2)11CH3 C=C Muscalure Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Strategy Muscalure is a C23 alkene The only functional group present is the ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a synthesis of (R)-CH3CHN3CH2CH3, starting from (R)-2-chlorobutane.
-
Propose a synthesis of bicyclohexylidene, starting from Cyclohexanone as the only source of carbon. Bicyclohexylidene
-
Beginning with acetylene and any alkyl halides needed, how would you synthesize the following compounds? (a) Decane (b) 2, 2-Dimethylhexane (c) Hexanal (d) 2-Heptanone
-
After attending a seminar on measuring customer profitability, Mason Ford decided to examine Olson Optics' customers to determine if the company truly knew how profitable its customers were. Olson...
-
The Calcio Coal Company produces coal at four mines and ships it to four power plants (P1-P4). The cost per ton of producing coal and the production capacity (in tons) for each mine are known. The...
-
The mean age at which females marry is 24.6. The standard deviation is 3.2 years. Find the corresponding z score for each. a. 27 b. 22 c. 31 d. 18 e. 26
-
Let \(D\) denote the event that you have the illness, and let \(S\) denote the event that the test signals positive. The probability requested can be denoted as \(P(D \mid S)\). The probability that...
-
If we can get that new robot to combine with our other automated equipment, well have a complete flexible manufacturing system (FMS) in place in our Northridge plant, said Hal Swain, production...
-
What impacts do you foresee the results of this survey will have on tourism in Canada (e.g. product development, consumption behavior, tourism marketing practice, etc.) (3 marks)? Assuming the role...
-
You work in a company that manufactures packed meals for school students. You want to sell your product to a big private school in your district. Write a sales letter to the school headmaster to...
-
How would you prepare Cyclodecyne starting from acetylene and any alkyl halide needed?
-
Compound A (C9H12) absorbed 3 equivalents of H2 on catalytic reduction over a palladium catalyst to give L (C9H18). On ozonolysis, compound A gave, among other things, a ketone that was identified as...
-
a. What is a catalyst? b. List the properties of catalysts. c. Name the catalyst used in the following processes: (i) The Contact process (ii) The Haber process (iii) The hydrogenation of unsaturated...
-
We sign a contract that calls for a down payment of 3 , 0 0 0 and for payments of 2 5 0 at the end of each month for 6 years. Money is worth 1 2 % compounded monthly. If we miss the first 8...
-
Suppose you are the financial manager of a firm considering the following five projects (expand below to see the five projects). Five Projects Under Construction Five Projects Under Consideration...
-
A 19-year bond with a 11.8 percent semiannual coupon and a $1,000 face value has a nominal yield to maturity of 9.9 percent. The bond currently sells for $1,161.31. The bond, which may be called...
-
Assume that ABC company wants to acquire a new equipment at an initial price of $ 1 0 0 0 with 1 0 years useful life. The equipment's value will be depreciated on a straight - line basis down to a...
-
What is the current yield for a bond with a current price of $1,200, a face value of $1,600, and a coupon rate of 5%? Round your answer to the nearest tenth.
-
Ethanol, a fuel, is made from corn. Ethanol production increased 8.5 times from 2000 to 2015 (www.ethanolrfa.org). Show the effect of this increased use of corn for producing ethanol on the price of...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Calculate the molarity of each solution. a. 0.38 mol of LiNO 3 in 6.14 L of solution b. 72.8 g C 2 H 6 O in 2.34 L of solution c. 12.87 mg KI in 112.4 mL of solution
-
The following compounds can all react as acids. (a) For each compound, show its conjugate base. Show any resonance forms if applicable. (b) Rank the conjugate bases in the order you would predict,...
-
In each reaction, label the reactants as Lewis acids (electrophiles) or Lewis bases (nucleophiles). Use curved arrows to show the movement of electron pairs in the reactions. Draw in any nonbonding...
-
In 1934, Edward A. Doisy of Washington University extracted 3000 lb of hog ovaries to isolate a few milligrams of pure estradiol, a potent female hormone. Doisy burned 5.00 mg of this precious sample...
-
(1) Given a soil with a moist unit weight of 107.7 pcf, a moisture content of 18.0 % and a specific gravity of 2.72, fill in all the blanks on the phase diagram. Also, find the void ratio, porosity,...
-
What are the importance of geotechnical engineering in building construction and civil engineering ?
-
Part #3 Consolidation test results of a settlement versus time on a soil sample for an increment of 200 kPa vertical stress is shown in the following table. Determine the value of the Coefficient of...

Study smarter with the SolutionInn App


