Question: The two peaks near m/z 31.00 in Figure 21-9 differ in mass by 0.010 Da. Estimate the resolving power of the spectrometer from the expression
The two peaks near m/z 31.00 in Figure 21-9 differ in mass by 0.010 Da. Estimate the resolving power of the spectrometer from the expression m/∆m without making any measurements in the figure.
Figure 21-9
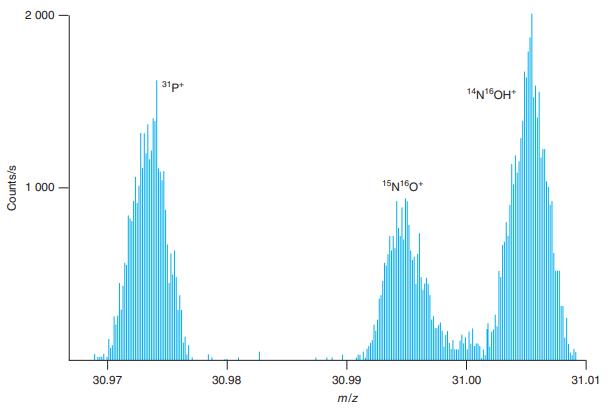
2 000 31p 14N1OH* 1 000 15N10* 30.97 30.98 30.99 31.00 31.01 m/z Counts/s
Step by Step Solution
★★★★★
3.49 Rating (166 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The overlap at the base of ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
878-E-C-E-E-C (2264).docx
120 KBs Word File


