Trans-Decalin is more stable than its cis isomer, but cis-bicyclo [4.1.0] heptane is more stable than its
Question:
Trans-Decalin is more stable than its cis isomer, but cis-bicyclo [4.1.0] heptane is more stable than its transisomer.Explain.
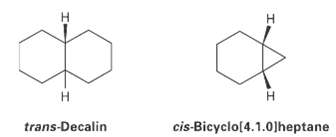
Transcribed Image Text:
trans-Decalin cis-Bicyclo[4.1.0]heptane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
Build models to see the stability difference between the two 410 rin...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Trans-2-Buterw is more stable than cic-2-hutene by only 4kJ/mol, but trans-2, 2, 5; 5-tetramethyl-3-hexene is more stable than its cis isomer by 3kJ/mol. Explain.
-
1-Methylcyclohexene is more stable than methylene-cyclohexane (A, in the margin), but methylene-cyclopropane (B) is more stable than 1-methyl-cyclopropene. Explain.
-
If a natural disaster, such as the 2010 drought in Russia, hits food production, use supply and demand analysis to figure out how this affects consumers and producers. Does everyone lose or are some...
-
1. What are the ways that Shelby and Mark might earn money from stock investments? What are the risks involved? 2. If Shelly and Mark want to use the Internet to evaluate stocks, what are four Web...
-
Determine the resultant internal loadings on the cross section at point D. 1.25 kN/m 1.5 m '0.5 m' 0.5 m'0.5 m
-
Analyze the housing price data in Table B. 4 for multicollinearity. Use the variance inflation factors and the condition number of \(\mathbf{X}^{\prime} \mathbf{X}\). y X1 X2 X3 X4 X5 X6 X7 Xg 25.9...
-
Desrosiers Ltd. had the following long-term receivable account balances at December 31, 2016: Notes receivable .................................................. $1,800,000 Notes receivable-Employees...
-
Calculate the current in each resistor in the following Network. What is the Potential difference voltage across the 50 resistor? 50 10 10 V 40 30
-
Discuss the meaning and elements of a valid contract under the Contracts Act 1950. Evaluate the principles related to the transfer of title under the Sale of Goods Act 1957. Recommend the amendments...
-
Using molecular models as well as structural drawings explain why trans-ans decalin is rigid and cannot ring-flip, whereas cis-decalin cans easily ring-flip.
-
Myo-lnositol, one of the isomers of 1, 2, 3, 4, 5, 6-hexahydroxvcyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation ofmyo-inositol. . " ....
-
Par Corporation paid $3,000,000 for an 80 percent interest in Son Corporation on January 1, 2011, when the book values and fair values of Son's assets and liabilities were as follows (in thousands):...
-
Late Adulthood COLLAPSE your presentation is not different from mine. I talk about changes in women's life and changes that occur in men's life at the end I present changes in cognition. i believe...
-
What information do you gain when you attend an annual conference of either the Mississippi Association of School Administrators, the Mississippi Association of High School Principals, the...
-
Explain the basic provisions of the federal FMLA (Family Medical Leave Act) - benefits, who is covered, requirements for companies to offer this benefit, etc. - Explain reasons why a federally...
-
your own point of view of what you believe are the pros/cons for utilizing a team based pay structure in an organization. Be as specific as you can of the plus and minus factors that would drive the...
-
As the first part of your response, describe in your own words (approximately 150-200 words) what research is and explain if your study will involve human subjects. Part 2: Research Ethics The...
-
A given coin has probability p = 0.37 of heads. What is the probability of tails?
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Compare the critical temperatures of NH 3 and N 2 (Table 12.6). Which gas has the stronger intermolecular forces? Table 12.6 TABLE 12.6 Some Critical Temperatures, T, and Critical Pressures, P...
-
From each set of resonance structures that follow, designate the one that would contribute most to the hybrid and explain your choice: (a) (b) (c) (d) (e) (f) HO HO
-
Give the structures of the products that would be formed when 1,3-butadiene reacts with each of the following: (a) (b) (c) (d) OMe OMe CN CN
-
Cyclopentadiene undergoes a Diels-Alder reaction with ethene at 160-180(C. Write the structure of the product of this reaction.
-
Wanda is reviewing her tax returns from the previous year and is shocked at how much tax she paid the government. She had good income but had to pay a large number of self-employment taxes on top of...
-
Katies Cleaning Service has cleaning contracts for 15 apartments, 45 family homes, and 25 office buildings. She estimates that an apartment takes 4 hours to clean, a home takes 6 hours to clean, and...
-
1. Quikpak sells returnable containers to major food processors. The price received for the containers is 2 per unit. Of this amount 1.25 is profit contribution. Quikpak is considering an attempt to...

Study smarter with the SolutionInn App


