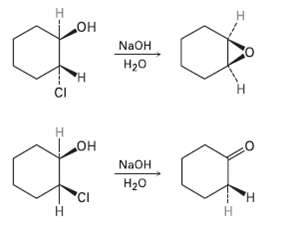
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the
Question:
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the same conditions yields Cyclohexanone. Propose mechanisms for both reactions, and explain why the different results areobtained.
Transcribed Image Text:
н но° N2OH H20 н но NAOH Н2о 'CI Н Н Н т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
H OH trans2Chloro cyclohexanol ring flip CI H OH cr H H 12Epoxyeyelohexane H cis2Chl...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose mechanisms for the reactions shown in Problems 22-62 parts (a) and (b) and 22-63 parts (a) and (b). In problem 22-62 (a) (b) In problem 22-63 (a) (b) CH3 TOH CH CH2-C-H CH CH OCH3 CH3OH CH3...
-
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
-
Propose reasonable fragmentation mechanisms that explain why The EI mass spectrum of benzoic acid shows major peaks at m/z = 105 and m/z = 77.
-
The costs of achieving emission reductions in the future will depend greatly on the types of policies used to reduce emissions today. Explain.
-
Which of Porter's Five Forces did Apple address through the introduction of the iPhone and customer developed iPhone applications?
-
A food scientist wants to study whether quality differences exist between yogurt made from skim milk with and without the pre culture of a particular type of bacteria, called Psychrotrops (PC)....
-
Does a correlation between hours of work and nonlabor income measure the income effect?
-
"Bell Company acquires 80% of Demers Company for $500,000 on January 1, 2009. Demers reported common stock of $300,000 and retained earnings of $200,000 on that date. Equipment was undervalued by...
-
What are the challenges and strategies for implementing TPM in highly regulated industries, such as pharmaceuticals or aerospace? How can TPM contribute to compliance and quality assurance in these...
-
A robot is used to prepare cases of peanut butter for shipment. As the 12 cases are being loaded, two of the cases are dropped. Before the operator can isolate the cases, they are mixed in with the...
-
Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason...
-
Ethers undergo an acid-catalyzed cleavage reaction when treated with the Lewis acid BBr3 at room temperature. Propose a mechanism for thereaction. + CHBr CH 1. r 2. H20
-
A 120-V ac voltage source is connected across a 2.0-F capacitor. Find the current to the capacitor if the frequency of the source is (a) 60 Hz and (b) 60 kHz. (c) What is the power loss in the...
-
Elle was 65 as of January 1st 2022. Her RRIF was established in 2020. On January 1, 2022, Elle's RRIF contained assets valued at $210,000, however, due to market fluctuations - her portfolio was...
-
What qualities and characteristics do you think a good leader must possess?
-
Pursuant to REIT Act of 2009, what item refers to the instruments or investment that are highly liquid and marketable and are considered good as cash as determined in a accordance with the rules and...
-
1.Does high income guarantee financial security? Why or why not? 2.How can people get more out of their money?
-
How portfolios can be used to reduce the risk exposure of investors but are unlikely to be able to eliminate risk exposure entirely?
-
Define the two major types of sampling strategies .
-
Classify each of the following activities as proper or prohibited under the various consumer statutes you have studied. a. Calling a hospital room to talk to a debtor who is a patient there. b....
-
Hydrogen has an auto-ignition temperature of 853 K; that is, hydrogen will ignite spontaneously at that temperature if exposed to oxygen. Hydrogen is to be adiabatically and reversibly compressed...
-
Write bond-line structural formulas for (a) Two primary alcohols, (b) A secondary alcohol, and (c) A tertiary alcohol-all having the molecular formula C4H10O.
-
One way of naming alcohols is to name the alkyl group that is attached to the -OH and add the word alcohol. Write bond-line formulas for (a) Propyl alcohol and (b) Isopropyl alcohol.
-
One way of naming ethers is to name the two alkyl groups attached to the oxygen atom in alphabetical order and add the word ether. If the two alkyl groups are the same, we use the prefix di-, for...
-
Superior Micro Products uses the weighted - average method in its process costing system. Data for the Assembly Department for May appear below: Materials Labor Overhead Work in process, May 1 $ 1 9...
-
Vista Distributors purchases inventory in crates of merchandise. Assume the company began July with an inventory of 30 units that cost $300 each. During the month, the company purchased and sold...
-
Blue Company changed depreciation methods in 2025 from double-declining balance to straight-line. Depreciation prior to 2025 under double-declining-balance was 592,400, whereas straight-line...

Study smarter with the SolutionInn App


