Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest
Question:
Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest a mechanism, using curved arrows to show electronmovement.
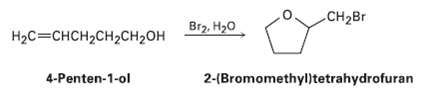
Transcribed Image Text:
CH2B Br2, H20 Нас%3 снсH2сH2сH20H 4-Penten-1-ol 2-(Bromomethyl)tetrahydrofuran
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Br HCCH2 Br 1 HC CH2 HCOH HC CH formation of cyclic bromonium io...View the full answer

Answered By

Tamil Elakkiya Rajendran
I'm currently involved in the research in the field of Biothermodynamics, Metabolic pathway analysis and computational Biology. I always prefer to share my knowledge whatever I have learnt through my degree whenever time permits.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use curved arrows to show how calcium carbide reacts with water to give acetylene.
-
Use curved arrows to show electron movement in each acid-base reaction in Problem 27.
-
Use curved arrows to show the electron movement in each reaction in Problem 33.
-
Find RL for maximum power transfer and the maximum power that can be transferred to the load in the circuit in figure. 1 mA 1 kn 2 mA RL 0.5 kn 1 kn 2 mA +)6 V
-
A recent study showed that women who lived near a freeway had an unusually high rate of rheumatoid arthritis. Sarah said, "They should move away from freeways." Is there a fallacy in Sarah's...
-
Reconsider the data in the previous question about number of hours spent studying. a. Find the median number of study hours for both males and females. What do these numbers tell us about the two...
-
Explain what the following accounting terms mean: a. Cost principle b. Conservatism
-
Decide which class of shares of GLEF is best for the investor if he plans to liquidate his investment toward the end of a. Year 1 b. Year 3 c. Year 5 d. Year 15
-
The client's contract called for MSI to conduct 1000 interviews under the following quota guidelines: 1. Interview at least 400 households with children. 2. Interview at least 400 households without...
-
The Britts & Straggon company manufactures small engines at three different plants. From the plants, the engines are transported to two different warehouse facilities before being distributed to...
-
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction: Co,CH3 C Hg(OAc)2 AcO-Hg
-
Hydroboration of 2-mnethyl-2-pentene at 25?C followed by oxidation with alkaline 11202 yields 2-methyl-3-pentanol, but hydroboration at 160?C followed by oxidation yields 4-methyl?1?pentanol. Suggest...
-
Suppose you are looking at two wire loops in the plane of the page as shown in Fig. 21-64. When switch S is closed in the left-hand coil, (a) What is the direction of the induced current in the other...
-
How does the supremacy clause of the constitution affect environmental law?
-
The ABC Company sells widgets at $ 9 each; variable unit cost is $ 6 , and fixed cost is $ 6 0 , 0 0 0 per year. What is the break - even quantity point? How many units must the company sell per year...
-
Wilson Properties is facing foreclosure on an apartment complex in Dallas, TX that was pur- chased six years ago. The property is financed with a $20 million nonrecourse loan, a tax basis of $10...
-
During 2022 the management of Capri Limited considered the acquisition a new machine for purchase and installation during the second quarter of 2024 with a desired rate of return of 15%. The machine...
-
List and describe at least three of the major U.S. environmental laws of the past 30 years.
-
Use the data in Table 1.4 to make a histogram of the U.S. dollar prices of a Big Mac in these 20 countries. Use these intervals for the prices: 12, 23, 34, 45, 58. United States Argentina Australia...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
The species NO 2 , NO 2 + , and NO 2 - in which N is the central atom have very different bond angles. Predict what these bond angles might be with respect to the ideal angles and justify your...
-
What products are formed when gach of the following ethers reacts with concenffated aqueous HI? 2-ethoxy-2,3-dimethylbutane
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


