What fragments might you expect in the mass spectra of the followingcompounds? (c) (b)
Question:
What fragments might you expect in the mass spectra of the followingcompounds?
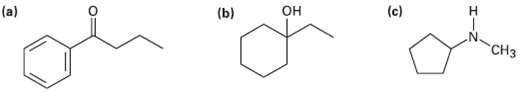
Transcribed Image Text:
(c) (b) он н "СНз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
a This ketone shows mass spectrum fragments that are due to alpha cleavage and ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
What kind of returns might you expect in the stock market? One way to measure how the stock market has performed is to examine the rate of return of the S&P 500 Index. To see historical prices of the...
-
What changes would you expect in the standard deviation for a portfolio of between 4 and 10 stocks, between 10 and 20 stocks, and between 50 and 100 stocks?
-
Shock Electronics sells portable heaters for $35 per unit, and the variable cost to produce them is $22. Mr. Amps estimates that the fixed costs are $97,500. a. Compute the break-even point in units....
-
It has been said that Porter's five-force analysis turns antitrust law on its head. What do you think this means?
-
Sample size is reported in APA format for a ingle-sample t test by reporting ____.
-
How does one distinguish between negligence and malpractice?
-
Sublime Sandals Company was formed on January 1, 2014, and is preparing its annual financial statements dated December 31, 2014. Ending inventory information about the four major items stocked for...
-
1. Why is the name of the Industry Standard used to provide a uniform basis for the estimating of building works? Answer: 2. Briefly explain the meaning of 'Request for Tender' in relation to gaining...
-
Owen Company manufactures bicycles and tricycles. For both products, materials are added at the beginning of the production process, and conversion costs are incurred uniformly. Owen Company uses the...
-
Assume that you are in a laboratory carrying out the catalytic hydrogenation of cyclohexane to cyclohexane. How could you use a mass spectrometer to determine when the reaction is finished?
-
How might you use IR spectroscopy to distinguish among the three isomers 1 -butyne, 1, 3-hutadiene, arid 2-butyne?
-
The generation of steam from the heat produced in the reformer is essential for economic viability of this energy-intensive operation. a. At what rate (kg/h) is superheated steam at 4.8 MPa and 100C...
-
a feasible study, can you provide specific examples of how theory has been or could be applied in the context of ABA professional recognition? This would help illustrate the practical application of...
-
Hello Dr. Jorgensen and Classmates, After watching the video by Dr. Laurence Steinberg, Professor of Psychology at Temple University, I found that there were a lot of good and relevant points that...
-
What has changed about the way you think about psychology after taking a psychology class? Why? How can you understand events that happen in your life through the lens of psychology (give an...
-
What are the changes in values, principles, and subject matter that cognitive psychology brought to the field of psychology?
-
A sample of gas occupies a volume of 260 ml when the temperature is 85C and the pressure is 750mm Hg. Both the temperature and pressure were changed causing the volume occupied by the gas to become...
-
What a difference a single outlier can make! The data set in Table 7.7 presents a collection of ordered pairs \((x, y)\). a. Using Minitab, draw a scatterplot and run a simple linear regression...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
Find the mass of urea (CH 4 N 2 O) needed to prepare 50.0 g of a solution in water in which the mole fraction of urea is 0.0770.
-
Predict the products of the following reactions. ether hexane + 2 Li hexane + 2 Li
-
Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde. (a) (b) (c) CH,OH CH,OH
-
Show two ways you could synthesize each of the following secondary alcohols by adding an appropriate Grignard reagent to an aldehyde. (a) (b) (c)
-
Factory overhead of $44,700 consists of Indirect labor of $21,900, Depreciation expense-Factory of $16,900, and Factory utilities of $5,900. a. Compute total manufacturing costs. b. Prepare a...
-
In Giannakakos et al. (2016), titled Using Video Modeling with voiceover Instruction plus feedback to train staff to implement direct teaching procedures, why do you think collecting data on the...
-
Presidio, Inc., produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidios cost data table. 2. Calculate Presidios contribution margin ratio and...

Study smarter with the SolutionInn App


