What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H.
Question:
What kind of hydribization do you expect for each carbon atom in the followingmolecules?
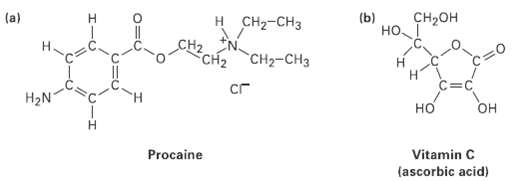
CH-он (b) H. н Cна-сHз (a) но 1 "CH-CHз CH2 C=C сг H2N H. но он Procaine Vitamin C (ascorbic acid)
Step by Step Answer:
a HN St H 302 H sp3 sp CC2 H ...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Review Examples 4 and 6 in the text. In both examples, the taxpayer's AGI is $129,400 even though in Example 6 there is $700 of nonrecaptured 1231 loss from 2015. Explain why the two AGI amounts are...
-
How are financial models superior to other screening models? How are they inferior?
-
This exercise explores the sensitivity of \(\log\)-optimality to the rebalancing frequency. Consider a market consisting of a risk-free asset with zero rate of interest and a stock that over 1 year...
-
The market value of the equity of Thompson, Inc., is $580,000. The balance sheet shows $35,000 in cash and $190,000 in debt, while the income statement has EBIT of $91,000 and a total of $135,000 in...
-
Think of a process--it could be as simple as making a peanut butter and jelly sandwich, or changing a light bulb. Using Gliffy, draw a flowchart for your process. What were some of your likes and...
-
Action Quest Games records adjusting entries on an annual basis. The company has the following information available on accruals that must be recorded for the year ended December 31, 2014: 1. Action...
-
Potassium methoxide, KOCH3, contains both covalent and ionic bonds. Which do you think is which?
-
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom. . Pyridoxal...
-
Alice purchases a rental house on June 22, 2020, for a cost of $174,000. Of this amount, $100,000 is considered to be allocable to the cost of the home, with the remaining $74,000 allocable to the...
-
Your branch target buffer (and branch history buffer) each have 8 slots. Your CPU executes the following code fragment. Assume that prediction takes place in the Fetch cycle, and that the result of a...
-
A 16-year-old girls father sues her fast-food employer when he discovers that a 29-year-old supervisor seduced and impregnated his daughter. What type of place are you running here, anyway? John...
-
Describe an ethical philosophy of leadership. This paper must include at least three sources and include key ethical principles, describe how this leader encourages others to act ethically and make...
-
a) Organizations are most vulnerable when they are at the peak of their success. It is for that reason even if it works, it must be fixed. Why is this so? b) As the Chief Executive of XYZ company,...
-
Richard purchased a 2010 hummer for $39,905. he made a down payment of $15,000 and paid $614 monthly for 4 years. find the APR Search instead for richard purchased a 2010 hummer for $39,905. he made...
-
Should the criminal law punish a person whose mere carelessness, as opposed to willfulness, causes harm to another?
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure and fugacity of nitrogen as a function of specific volume at the two temperatures a. 110 K b. 150 K
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
How may price floors and ceilings destroy market equilibrium? How does the shape of the indicator line on the graph show diminishing returns? Monthly rent A Price Ceiling on Rent B $900 $800...
-
Provide a comprehensive analysis of the regulatory frameworks governing financial institutions, delineating the intricate interplay between prudential supervision, market conduct standards, and...
-
How does the forum meet organisational objectives? What vocabulary, tone, structure and style suits the audience? How will I: build trust develop positive working relationships ...

Study smarter with the SolutionInn App


