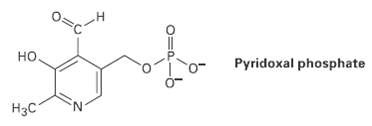
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions.
Question:
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom.
Transcribed Image Text:
но. Pyridoxal phosphate Нас 0=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
sp3 sp2 H3C H sp2 sp2 sp2 sp2 5p3 Pyridoxa...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the molecular structure and the bond angles for each molecule or ion in Exercises 57, 58, and 60.
-
Predict the molecular structure and the bond angles for each of the following. a. SeO3 b. SeO2 c. PCl3 d. SCl2 e. SiF4
-
Predict the molecular structure and the bond angles for each of the following. (See Exercises 89 and 90.) a. XeCl2 b. ICl3 c. TeF4 d. PCl5
-
In the Akerlof example, the individuals are treated as indifferent to risk. What would you expect to see in these markets if individuals wanted to avoid risk? What if there were some risk lovers?
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
Construct a network activity diagram based on the following information: Activity Preceding activities A - B - C A D B, C E B F D G E H F I G, H
-
You are managing a pension fund with a goal of maximizing the long-term growth rate. There are three assets available. Asset 1 has a risk-free return of 5%. Assets 2 and 3 each are driven by...
-
Using the data in BE4-6, journalize and post the entry on July 1 and the adjusting entry on December 31 for Craig Insurance Co. Craig uses the accounts Unearned Service Revenue and Service Revenue.
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor - hours. At the beginning of the year, it estimated that 2 5 , 0 0 0 direct labor - hours would...
-
How might have the individual difference variables have influenced the personality of this individual? Also define the cognitive variable including specific personality attributes and examples. Jose...
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
In an ionic crystal lattice each cation will be attracted by anions next to it and repulsed by cations near it. Consequently the coulomb potential leading to the lattice energy depends on the type of...
-
Go to the Internet site of the state courts in your state. (See Appendix D.) Find the court rules of any civil court in your state that hears divorce cases. Quote from any court rule that uses the...
-
How do evolutionary theories of strategy illuminate the adaptive processes by which organizations explore new opportunities, exploit existing capabilities, and continuously renew their competitive...
-
What are the implications of open innovation strategies for reshaping traditional approaches to value creation and competitive positioning within ecosystems of collaborators and competitors?
-
How do emergent properties of complex adaptive systems influence the formulation and execution of organizational strategy in dynamic environments?
-
6. What are some advantages and disadvantages of using a single I/O interrupt queue? 7. What are some advantages and disadvantages of using multiple I/O interrupt queues? Job A B C D E F| Arrival...
-
Is there a justification for the criminal law not punishing a person for failing to act when there is a clear moral duty to act?
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Can a fluid obeying the virial equation of state have a vapor-liquid transition?
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
If a set of m+1 data points (x,y) are to be represent by a least squares fit of y=c logx derive an expression for c in terms of x and y.
-
5 If 0 = then 4 cos(0) = sin(0) =
-
Determine the reactions of the given beam using slope deflection method. P = 25 kN, P2 = 25 kN, and w = 25 kN/m P1 P2 2m B 2m C 4m 21 Numerical answer in 4 decimal places and positive values only: RA...

Study smarter with the SolutionInn App


