What product(s) would you expect to obtain from the followingreactions? (b) r. 1. HNO3, H2SO4 2. Fe,
Question:
What product(s) would you expect to obtain from the followingreactions?
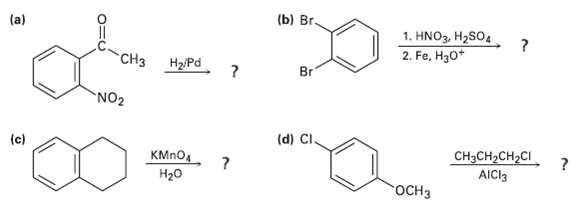
Transcribed Image Text:
(b) Вrг. 1. HNO3, H2SO4 2. Fe, H30* "CНз Br H2/Pd NO2 (d) CI. (c) CH3CH2CH2CI КMпO Н2о AICI3 гоСНз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
3 b Br CH3 NO oEthylaniline Catalytic hydrogenation reduces bo...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What stereoisomers would you expect to obtain from each of the following reactions? a. b. c. d. e. f. CH,CH CHs Br2 CH2C2 CH3 CH2CH3 CHCH CH Pt/c H3C CH2CH3 Br2 CH2Cl2 CH3 CH CH2C2 CH CH2CH H2 Pt/C...
-
What products would you expect to obtain from each of the following crossed Claisen condensations? (a) (b) Ethyl propanoate+ (1) NaOEt (2) H,o yl oxalate (1) NaOEt Ethyl acetate ethyl formate (2) H,O
-
What products would you expect to obtain from the complete hydrolysis of NAD? See page 547 for its structure.
-
As a bakery business continues to grow, cash flow has become more of a concern. The board of directors would like to maintain the market share price, so a discussion ensues about issuing a stock...
-
Mary Marble took out a homeowner's insurance policy on her residence. One of the policy provisions listed the insured's duties with respect to a loss. This provision required the policyholder to give...
-
Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed by an epoxide, followed by heating in aqueous acid: Using this process, identify what...
-
The probability that a wafer contains a large particle of contamination is 0.01. If it is assumed that the wafers are independent, what is the probability that exactly 125 wafers need to be analyzed...
-
Consistent Company has been paying regular quarterly dividends of $1.50 and wants to pay the same amount in the third quarter of 2011. Given the following information, (1) What is the total amount...
-
Vulcan Company's contribution format Income statement for June is as follows: Vulcan Company Inco Stat For the Month Ende June 30 Sales Variable expens Contribution margin Fixed expenses Net...
-
Mini-Case Study: The Back to School Crunch at Global Green Books Publishing Global Green Books Publishing is a successful printing and publishing company. Just two years old, it has taken on a great...
-
Rank the following aromatic compounds in the expected order of their reactivity toward FriedelCrafts alkylation. Which compounds are un-reactive? (a) Bromobenzene (b) Toluene (c) Phenol (d) Aniline...
-
Predict the major product(s) of the followingreactions: (b) (a) CH3CH2COCI AICI3 CH3CH2CI AICI3 (c) (d) N(CH2CH3}2 CO2H HNO3 so3 H2SO4 H2SO,
-
Figure Q18.15 shows a photograph of droplets of water suspended from the stem of a plant in a garden, with a flower behind. There appear to be small flowers in each of the droplets; explain what is...
-
What is an example of a promotional message that went truly viral? How can you tell if it was truly viral or whether it was seeded through paid online marketing and a public relations strategy? An...
-
B began business 3 / 1 5 with a cash investment of 2 5 0 0 0 , The records show: Sales for the balance of the year 5 6 0 0 0 , accounts receivable 1 2 / 3 1 3 0 0 0 0 , accounts payable 1 2 / 3 1 2 0...
-
Why can't you merge an income account with an expense account in the Chart of Accounts?
-
XYZ Corporation, a multinational manufacturing company, is considering a significant investment in a new production facility. The capital budget for this project involves substantial financial...
-
LensWorks manufactures reading glasses in two styles. They budgeted a volume of 15,000 units for style A, at a selling price of $10 each. The variable costs per unit were budgeted at $2 per unit. The...
-
Should environmental crimes that carry major penalties be strict liability offenses, or should prosecutors be required to prove that a defendant knowingly or willfully committed an offense?
-
Represent each of the following combination of units in the correct SI form using an appropriate prefix: (a) m/ms, (b) k m, (c) k s /mg, and (d) k m N.
-
Redo Problem 5.2 using Aspen Plus. Problem 5.2 t is desired to improve the thermal efficiency of the Rankine power generation cycle. Two possibilities have been suggested. One is to increase the...
-
Show how this synthesis might beaccomplished: Br CH3 CH3 HO from "CN -
-
What is wrong with these reactions explain. CI + NaOCH3 OCH3 + NaCl a) + HBr Br . b) CH,OH + CH;0 OCH3 CI d)
-
What is wrong with these synthesesexplain. 1) NaNH2, NH, (1) C=CCH3 a) CH3C=CH 2) -Br CH;CH,-NH, I b) CH;CH,I + NH3 CI + Br OCH, CH, + CH,O d) . .. Br + HBr e) CH3 CH3 H,SO, f)
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


