Question: When 2-methylcyclohexanone is converted into an enamine, only one product is formed despite the fact that the starting ketone is unsymmetrical. Build molecular models of
When 2-methylcyclohexanone is converted into an enamine, only one product is formed despite the fact that the starting ketone is unsymmetrical. Build molecular models of the two possible products, and explain the fact that the sole product is the one with the double bond away from the methyl- substitutedcarbon.
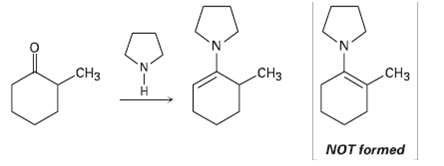
CH3 CH3 CH3 NOT formed
Step by Step Solution
3.43 Rating (162 Votes )
There are 3 Steps involved in it
CH CH3 Crowding between the methyl group and the py... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-C-R (52).docx
120 KBs Word File


