Question: Which of these compounds can form an intra molecular (within the same molecule) hydrogen bond between the hydrogen of the carboxylic acid and the oxygen
Which of these compounds can form an intra molecular (within the same molecule) hydrogen bond between the hydrogen of the carboxylic acid and the oxygen of the ether group? Explain.
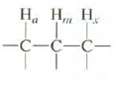
H, H, H -C-c-c- |
Step by Step Solution
★★★★★
3.31 Rating (177 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The tertbutyl group has a very large axial strain energy an... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
15-C-O-S (73).docx
120 KBs Word File


