Write the mechanism of the reaction shown in figure between coenzyme A and acetyl adenylate to give
Question:
Write the mechanism of the reaction shown in figure between coenzyme A and acetyl adenylate to give acetylCoA.
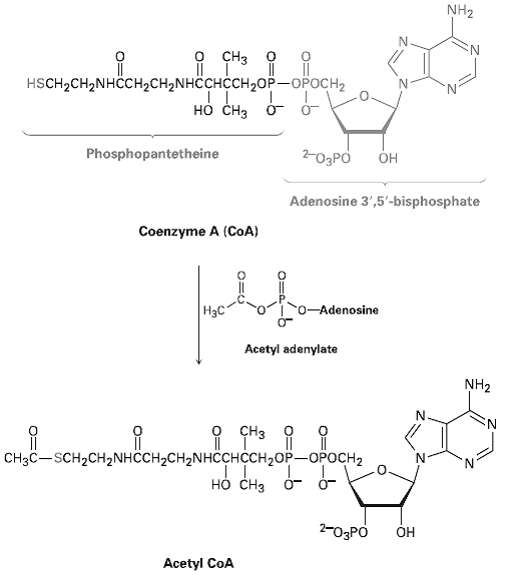
Transcribed Image Text:
NH2 оооне р CHз Д Т"3 HSCH-CH2NHCCH2CH2NHCCHCCH,0P — ОРОСНа но снз о- о- Phosphopantetheine 2-O3PO Он Adenosine 3',5'-bisphosphate Coenzyme A (CoA) го- Adenosine | Нас Acetyl adenylate NH2 о Cнз о П нонивенони ре. CH3C-SCH2CH2NHCCH2CH2NHCCHCH2OP-OPOCH2 но снз 2-03PO ОН Acetyl CoA
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Even though the entire molecule of coenzyme A is biologically important we are concer...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
(a) According to the mechanism of the reaction shown in Eq. 10.24, what would be the absolute configuration of the alkyl chloride obtained from the reaction of thionyl chloride. With (S)...
-
Write the mechanism of the reaction just shown between 3, 4, 5-trimethoxybenzoyl chloride and morpholine to form trimetozine. Use curved arrows to show the electron flow in each step.
-
Write the mechanism of the reaction between p-hydroxyaniline and acetic anhydride to prepare acetaminophen.
-
In the first case, the relationship between F and H is found to be inverse when the height decrease the area will increase, and the force will increase too. Therefore, I need a logical and practical...
-
Watch Don McMillan's now famous YouTube hit "Life After Death by PowerPoint" from 2008 or the expanded version "Life After Death by PowerPoint 2010." Which specific PowerPoint ills is McMillan...
-
A storage tank (6 m 6 m 6 m) is filled with water. Determine the force on the bottom and on each side.
-
Find the interquartile range of the data set in Example 1. Are there any outliers? Data from Example 1 Each year in the U.S., automobile commuters waste fuel due to traffic congestion. The amounts...
-
Peter is a professor of mathematics at State University. His lifetime avocation has been sailing, and he owns an oceangoing sailing vessel. He plans to retire in five years and spend the remainder of...
-
Draw a Sankey Diagram of the energy flow to power an electric vehicle. Normalize the energy consumption of the vehicle to 1 ( that is , the width of the vehicle energy consumption flow will be 1...
-
E-Education is a new start-up that develops and markets MBA courses offered over the Internet. The company is currently located in Chicago and employs 150 people. Due to strong growth the company...
-
How would you use the reaction of an amide with LiAlH4 as the key step is going from bromo cyclohexane to (N, N-dimethylaminomethyl) cyclohexane? Write all the steps in the reactionsequence. Br...
-
Draw structures of the step-growth polymers you would expect to obtain from the followingreactions: Base (a) BrCH-CH2CH2Br + 2H2CH2H H2SO, catalyst () -H2 + -CICH2/6CO2H (c) l H2N(CH216NH2 +...
-
What do you mean by database metadata? How do you get them?
-
During 2023, Gescan Electric Inc. has performed well, so the board of directors decided to pay dividends. On November 30, 2023, the company declared cash dividends of $147,000, which will be paid out...
-
Describe three types of heredity-environment correlations, and give an example of each .
-
Draw all the isomers of C5H0. Clearly show stereochemistry if stereoisomers are possible. Step 1: Start by drawing all the isomers with double bonds. . Consider constitutional isomers, then...
-
EFG declared a property dividend. The dividend consisted of 10,000 common shares of its investment in TUV Company. The shares had originally been purchased at $4 per share and had a $1 par. The value...
-
Explain the conditions for implementing cooperation in the use of state/regional goods by the provisions of Government Regulation 6 of 2006 & Government Regulation 38 of 2008.
-
The Police Department is interested in determining the e ect of alcohol consumption on driving performance. Twelve male drivers of similar weight, age, and driving experience were randomly assigned...
-
Economic feasibility is an important guideline in designing cost accounting systems. Do you agree? Explain.
-
Sketch the bonding and antibonding molecular orbitals that result from linear combinations of the 2p x atomic orbitals in a homonuclear diatomic molecule. (The 2p x orbitals are those whose lobes are...
-
Give a single structure that fits the data provided. A compound C5H9C13 has three NMR absorptions at 1.99, 4.31, and 8 6.55 with relative integrals of 6:2:1, respectively.
-
Give a single structure that fits the data provided. A compound C5H9C13 has three NMR absorptions at 1.99, 4.31, and 8 6.55 with relative integrals of 6:2:1, respectively.
-
Strange results in the undergraduate organic laboratory have led to the admission by a teaching assistant, Thumbs Throckmorton, that he has accidentally mixed some terf-butyl bromide with the methyl...
-
Martin Company applies manufacturing overhead based on direct labor hours. Information concerning manufacturing overhead and labor for the year is as follows: Actual manufacturing overhead Estimated...
-
$ 719,000 280,000 439,000 150,400 Joyner Company's income statement for Year 2 follows: Sales Cost of goods sold Gross margin Selling and administrative expenses Net operating income Nonoperating...
-
FIFO and LIFO costs under perpetual inventory system The following units of an item were available for sale during the year: Beginning inventory 22 units at $44 Sale 18 units at $62 First purchase 18...

Study smarter with the SolutionInn App


