How would you use the reaction of an amide with LiAlH4 as the key step is going
Question:
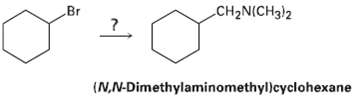
How would you use the reaction of an amide with LiAlH4 as the key step is going from bromo cyclohexane to (N, N-dimethylaminomethyl) cyclohexane? Write all the steps in the reactionsequence.
Transcribed Image Text:
Br CH2N(CH3)2 (N,N-Dimethylaminomethyl)cyclohexane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Br Mg ether CHNCH32 1 LIAIH4 2 HO MgBr OC 1 CO ether 2 HO NCH32 CH32NH NaOH OC SOCI O...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you use the net marketing contribution for a company like Clorox to forecast net profit and earnings per share?
-
How would you use the graphic on this chapter's first page and to assess Apple's 2010 marketing ROS (27.5%) and marketing ROI (323%)?
-
How would you use the control variate approach to improve the estimate of the delta of an American option when the binomial tree approach is used?
-
(a) Employing the intercept technique, determine the average grain size for the steel specimen whose microstructure is shown in Figure (a); use at least seven straight-line segments. (b) Estimate the...
-
What is a customized résumé, and why should you have one?
-
A small amount of solvent is added to the ground water to change its electrolytic content. As a result, the contacting angle, 0, representing the adhesion between water and soil material, is...
-
Draw a box-and-whisker plot that represents the data set in Example 1. What do you observe? Data from Example 1 Each year in the U.S., automobile commuters waste fuel due to traffic congestion. The...
-
The Welch Company is considering three independent projects, each of which requires a $5 million investment. The estimated internal rate of return (IRR) and cost of capital for these projects are as...
-
A car runs on wheels of diameter 3 5 0 mm . The car is travelling at an average speed of 7 5 km / h , what is the angular velocity of the wheels?
-
FlyHigh Tech, an early-stage startup, reports to its investors using a balanced scorecard that is prepared at the end of each quarter. During the first and second quarters of the current year, the...
-
How would you convert N-ethylbenzamide to each of the following products? (a) Benzoic acid (b) Benzyl alcohol (c) C 6 H 5 CH 2 NHCH 2 CH 3
-
Write the mechanism of the reaction shown in figure between coenzyme A and acetyl adenylate to give acetylCoA. NH2 CH "3 HSCH-CH2NHCCH2CH2NHCCHCCH,0P - - Phosphopantetheine 2-O3PO Adenosine...
-
A typical household pays about $1200 a year on energy bills, and the U.S. Department of Energy estimates that 46 percent of this energy is used for heating and cooling, 15 percent for heating water,...
-
Amber, a hotel manager, was required to accept free lodging in the hotel in connection with her obligations as the hotel manager. This lodging is valued at $5,000 for the year. In addition to this...
-
What are the differences between the family in Malawi and the family in Cambodia ?
-
What ability did Engle assess using methods like the reading span task to identify individual variances ?
-
When setting up the trust book accounting for an Agency, what important basic requirements need to be considered (include at least three)?
-
Introduction: (roughly one page) Start with a "Hook" to draw the reader in and then explain in broad terms how the most recent recession began. II. Consequences of great recession (roughly one page)...
-
A researcher is investigating the relationship between fuel economy and driving speed. He makes six runs on a test track, each at a di erent speed, and measures the kilometers traveled on one liter...
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Draw an MO energy diagram and predict the bond order of Li 2 + and Li 2 . Do you expect these molecules to exist in the gas phase?
-
The ==C-H stretching absorption of 2-methyl- I-pentene is observed at 3090 cm-1. If the hydrogen were replaced by deuterium, at what wavenumber would the ==C-D stretching absorption be observed?...
-
Which of the following vibrations should be infrared-active and which should be infrared inactive (or nearly so)? (a) (b) (c) (CH, ),C-=0 C-O stretch C C stretch (CHC CI C-Cl stretch
-
What magnetic field at the proton would be required to cause "spin flipping" in an NMR experiment in which the frequency imposed on the sample is 900 MHz?
-
An all-equity firm is considering the purchase of a depreciable asset that costs $680,000 and would be fully depreciated over six years using the straight-line method. The asset is expected to...
-
A firm owns three department stores. Each store has a debt-equity ratio of 16 percent and makes interest payments of $44,000 at the end of each year. The cost of the firm's levered equity is 18...
-
Belgian Bite Corp. is considering a project that has the following cash flow data. What is the project's IRR? Year 0 1 2 3 Cash flows $1,100 $450 $470 $490

Study smarter with the SolutionInn App


