?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is
Question:
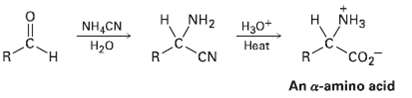
?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous acid. Propose a mechanism for the reaction.
Transcribed Image Text:
н NH2 H NH3 Нао* NH,CN Нао Heat CN co2 An a-amino acid R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
NH NH3 addition of NH3 COH H R NH3 H ...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following amines can be prepared by the Gabriel synthesis? Which ones cannot? Write equations showing the successful applications of this method. (a) Isobutylamine (b) tert-Butylamine...
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Dr. Thomas Bilski is a primary care physician with a practice that is currently serving 280 patients. Dr. Bilski would like to administer a survey to his patients to measure their satisfaction levels...
-
Using your team's answers to Activity 4, make the statements stronger by quantifying them (make up any numbers you need).
-
One of the procedures in the production of integrated circuits is the formation of a thin insulating layer of SiO2 on the surface of chips (see Figure 18.26). This is accomplished by oxidizing the...
-
(a) Describe briefly what is meant by 'an audit programme". (b) State the two stages in which an audit programme is developed.
-
At December 31, 2015, Ingleton Company reports the following results for the year: Cash sales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $1,025,000 Credit sales . . . . . . . . . . ....
-
Green Co. has a checking account at Red Bank and an interest-bearing savings account at Blue Bank. On December 31, Year 1, Green's bank records reflect the following information: Red Bank Bank...
-
1. Considering Yips globalisation framework (Figure 9.2), what drivers of internationalisation do you think were most important when Wanda entered the US market through its AMC and Legendary...
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
One of the reactions used in determining the sequence of nucleotides in a strand of DNA is reaction with hydrazine. Propose a mechanism for the following reaction, which occurs by an initial...
-
What impact does multiple geographical business locations have on the client acceptance decision?
-
Apply the methods in the ListIterator interface to write a Java program in NetBeans that creates a LinkedList of four elements of type string, namely: Java, C#, PHP and Python. The program should...
-
D Question 1 Define P(n) to be the assertion that: n(n+1) (2n+1) The first step in mathematical induction is to verify the base case. Determine what the base case should be. calculate its value, and...
-
n = 6 Type the program's output while n >= 0: print (n-2) nn 1
-
Draw a graph that shows marginal cost, average variable cost, and average total cost. Your curves only need to have the proper shape and relationship to the other curves; you do NOT need to plot your...
-
Consider the CAPM. The expected return market is 20%. The expected return on a stock with a beta of 1.2 is 22%. What is the risk free rate?
-
How much money can be withdrawn at the end of the investment period if a. $1,000 is invested at 8 percent/year compounded annually for 10 years? b. $5,000 is invested at 11 percent/year compounded...
-
Dawson Companys balance sheet information at the end of 2019 and 2020 is as follows: Additional information: The company did not issue any common stock during 2020. Required : Next Level Fill in the...
-
Define pH. What pH range is considered acidic? Basic? Neutral?
-
The structure of the nitro group (-NO2) is usually shown as Experiments show that the two nitrogen-oxygen bonds have the same length of 1.21 Ã. This length is intermediate between 1.36...
-
Draw all reasonable resonance structures for naphthalene (C10H8, Sec. 4.13) and rationalize the different bond lengths for the C-C bonds.
-
When naphthalene is hydrogenated, the heat released is about 80 kcal/mol. Using an isolated cyclohexene unit for comparison, estimate the resonance energy of naphthalene. Why is the resonance energy...
-
Describe the phenomenon in current accounting practices that has been dubbed the Earnings Game in the article by Justin Fox. Why do companies play this game? Based off this article:...
-
On January 1, 2018, Bark Manufacturing Company Ltd. purchased a machine for $27,500, and expects to use the machine a total of 32,000 hours over the next four years. Bark set the residual value on...
-
Covid and post Covid Economic Recovery for each of the papers you are required to state the following : 1. what was the study about 2. what were the findings 3. list the research gaps and suggest new...

Study smarter with the SolutionInn App


