Question: A 250 g block is dropped onto a relaxed vertical spring that has a spring constant of k = 2.5 N/cm (Figure).The block becomes attached
A 250 g block is dropped onto a relaxed vertical spring that has a spring constant of k = 2.5 N/cm (Figure).The block becomes attached to the spring and compresses the spring 12 cm before momentarily stopping. While the spring is being compressed, what work is done on the block by?(a) The gravitational force on it and(b) The spring force?(c) What is the speed of the block just before it hits the spring? (Assume that friction is negligible.)(d) If the speed at impact is doubled, what is the maximum compression of thespring?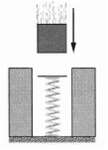
wwww
Step by Step Solution
3.43 Rating (166 Votes )
There are 3 Steps involved in it
a The compression of the spring is d 012 m The work done by the force of gravit... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
2-P-M-K-E (54).docx
120 KBs Word File


