A mixture of cyclohexane and cyclopentane is to be separated by liquid-liquid extraction at 25 o C
Question:
A mixture of cyclohexane and cyclopentane is to be separated by liquid-liquid extraction at 25oC with methanol. Phase equilibria for this system may be predicted by the NRTL or UNIQUAC equations. Calculate product rates and compositions and interstage flow rates and compositions for the conditions in figure with:
(a) N = 1 equilibrium stage.
(b) N = 2 equilibrium stages.
(c) N = 5 equilibrium stages.
(d) N = 10 equilibriumstages.
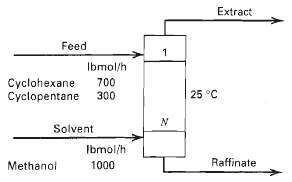
Extract Feed Ibmol/h Cyclohexane Cyclopentane 300 700 25 °C Solvent Ibmol/h Raffinate Methanol 1000
Step by Step Answer:
These data are fitted to the NRTL and UNIQUAC equations in LiquidLiquid Equilibrium Data Collection Ternary Systems Their fit to the simpler NRTL equa...View the full answer



Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Chemical Engineering questions
-
A mixture of methane and ethane is to be separated by distillation. Explain why water cannot be used as the coolant in the condenser. What would you choose as the coolant?
-
A mixture of ethylene and ethane is to be separated by distillation. Determine the maximum operating pressure of the column. What operating pressure would you suggest? Why?
-
The interval [0, 1] is to be separated into three pieces by making cuts at x and y. Let A(x, y) be the area of any nondegen-crate triangle that can be formed from these three pieces. Deter-mine the...
-
Joseph Kent started business on 1 July 2018 as a joiner making conservatories. His taxadjusted profits (before deduction of capital allowances) were as follows: Private use of both cars has been...
-
The basic accounting equation is Assets = Liabilities + Shareholders' Equity. Replacing words with dollar amounts, what is Eastplats's accounting equation at December 31, 2010?
-
The 2-kg collar is at rest in position A when the constant force P is applied as shown. Determine the speed of the collar as it passes position B if (a) P = 25 N (b) P = 40 N. The curved rod lies in...
-
Suppose that in the maintenance of a large medicalrecords file for insurance purposes the probability of an error in processing is 0.0010, the probability of an error in filing is 0.0009, the...
-
The Z-score bankruptcy prediction model uses balance sheet and income information to arrive at a Z-Score, which can be used to predict financial distress: EBIT is earnings before interest and taxes....
-
A lake with a fixed carrying capacity contains a certain fish population. The fish population in the lake has a growth rate that is proportional to its size when the population is very small relative...
-
Deepa Dalal opened a free-standing radiology clinic. She had anticipated that the costs for the radiological tests would be primarily fixed, but she found that costs increased with the number of...
-
Calculate the product compositions, stage temperatures, interstage flow rates and compositions, and reboiler duty for the re-boiled stripper shown in Figure10.40. Overhead vapor Feed 39.2F, 150 psia...
-
The liquid-liquid extractor in figure operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
While having lunch in the staff room, Jorge is obviously mad. He spent one morning taking a meticulous social history from a new client. The client, a man in his 20s, was pleasant and helpful. He...
-
Write a function which deletes first node from the singly circular linked list?
-
Implement C code for linear queue using linked list with a suitable example.
-
Show that along the optimal trajectory the quantity is a constant. Deduce from this that along the trajectory the quantity f (t) = y/x is a constant K, and express its value in terms of C, g, and . A...
-
What behaviors do you see the most from new leaders? Hands off, hero, or best friend?
-
What resistance do you think youll see in your next conversation? Is it within yourself or the other person?
-
In the disclosure of capitalized costs relating to oil and gas producing activities, which cost categories should not be included? a. Mineral interests in properties are not included. b. Support...
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
What is core capital? How do risk-adjusted assets differ from total assets?
-
Representative properties of small-pore silica gel are: d p = 24 A, p = 0:47, p = 1.09 g/cm 3 , and S g = 800 m 2 /g. (a) Are these values reasonably consistent? (b) If the adsorption capacity for...
-
In adsorption processes, what distinguishes purification from bulk separation?
-
Ruthven and Kaul [Ind. Eng. Chem. Res., 32, 20472052 (1993)] measured adsorption isotherms for a series of gaseous aromatic hydrocarbons on well-defined crystals of NaX zeolite over ranges of...
-
Jet Airways borrowed $1,000,000 for one year to improve its liquidity. Jet Airways paid back the principal and interest of $1,060,000 at the end of the year. What is the dollar amount of interest and...
-
if the cost of attending the university is $40,000 per year due at the beginning instead of the end of each year for four years, how much do you have to deposit in a lump sum to be able to pay for...
-
Financial Analysis Case This case can be assigned as a group activity. Additional instructions and material for this activity can be found on the Instructor Resource site and in WileyPLUS. Kenmare...

Study smarter with the SolutionInn App


