Calculate the product compositions, stage temperatures, interstage flow rates and compositions, and reboiler duty for the re-boiled
Question:
Calculate the product compositions, stage temperatures, interstage flow rates and compositions, and reboiler duty for the re-boiled stripper shown in Figure10.40.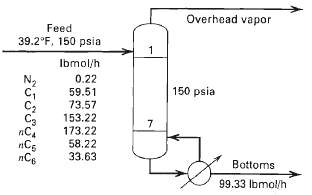
Transcribed Image Text:
Overhead vapor Feed 39.2°F, 150 psia Ibmol/h 0.22 N2 150 psia 59.51 73.57 153.22 173.22 58.22 33.63 Ca nCg Bottoms 99.33 Ibmol/h
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (15 reviews)
The calculations were made with the Tower model Insideout method of Chemcad wit...View the full answer

Answered By

HILLARY KIYAYI
I am a multi-skilled, reliable & talented Market analysis & Research Writer with a proven ability to produce Scholarly Papers, Reports, Research and Article Writing and much more. My ultimate quality is my English writing/verbal skill. That skill has proven to be the most valuable asset for project writing, Academic & Research writing, Proofreading, HR Management Writing, business, sales, and a variety of other opportunities.
4.80+
24+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Calculate product compositions, stage temperatures interstage vapor and liquid flow rates and compositions, reboiler duty, and condenser duty for the following distillation-column specifications Feed...
-
Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for an absorber having four equilibrium stages with the specifications in...
-
Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for the absorber shown inFigure. Lean gas Lean oil, 80F, 400 psia 250 Ibmol/h...
-
An individual has capital losses brought forward from previous years amounting to 4,800. Compute the individual's taxable gains for 2020-21 if total gains and losses for the year are as follows:...
-
Why should everyone study accounting whether they are going to be an accountant or not?
-
How does the taxation of a gain recognized when a U.S. shareholder sells stock in a CFC differ from that of a gain recognized when a U.S. shareholder sells stock in a non-CFC?
-
You have been assigned to the first examination of the accounts of The Chicago Company for the year ending March 31, 19X1. The accounts receivable were circularized at December \(31,19 X 0\) and at...
-
The Pear Computer Company just developed a totally revolutionary new personal computer. It estimates that it will take competitors at least two years to produce equivalent products. The demand...
-
1. The following information was obtained from Durand, Inc.: Advertising Costs $11,400 Indirect Labor 5,000 CEO's Salary 530,000 Direct Labor 59,000 Indirect Materials Used 5,500 Direct Materials...
-
Suppose you work for a multinational firm and are posted to Bogota, Colombia. After renting a house in a posh neighborhood, you hire a full-time maid to perform household chores, a common practice...
-
Determine product compositions, stage temperatures, interstage flow rates and compositions, and reboiler duty for the reboiled absorber shown in Figure 10.39. Repeat the calculations without the...
-
A mixture of cyclohexane and cyclopentane is to be separated by liquid-liquid extraction at 25 o C with methanol. Phase equilibria for this system may be predicted by the NRTL or UNIQUAC equations....
-
Rounding for the following matrix A find det A, what happens if you round off the fiven entries to(a) 5S,(b) 4S,(c) 3S,(d) 2S,(e) 1S?What is the practical implication of yourwork? A = 1/4 1 1/3 -1/9...
-
Report the results of a fitted regression equation in such a way that confidence intervals and hypothesis tests for the unknown coefficients can be constructed quickly and easily.
-
One end of a short aluminum rod is in a campfire and the other end is in a block of ice, as shown in Figure Q11.8. If \(100 \mathrm{~J}\) of energy are transferred from the fire to the rod, and if...
-
Let \(y=\) expenditure ( \(\$\) ) on food away from home per household member per month in the past quarter and \(x=\) monthly household income (in hundreds of dollars) during the past year. a. Using...
-
In an isolated system far from thermal equilibrium, as time passes, A. The total energy stays the same; the total entropy stays the same. B. The total energy decreases; the total entropy increases....
-
A fire piston-an impressive physics demonstration-ignites a fire without matches. The operation is shown in Figure Q11.18. A wad of cotton is placed at the bottom of a sealed syringe with a...
-
The shown partners have always shared profits and losses in the ratio: Holt 4; Stott 2: Young 1. From 1 January the assets were to be revalued as the profit sharing ratios are to be altered soon. The...
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
Who regulates money markets? Capital markets?
-
Why is it easy to measure the amount of adsorption of a pure gas, but difficult to measure adsorption of a pure liquid?
-
In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binarysolute data point. Use the mixture data below to obtain the best fit to...
-
What is meant by regeneration?
-
Compare and contrast different deadlock prevention techniques such as resource ordering, the "hold and wait" condition, and preemptive resource allocation. Which methods are most effective for...
-
The following information is taken from Aden Company's records: Product Group Units Cost/Unit Market/Unit A 1 700 $1.10 $0.90 B 1 250 1.50 1.55 C 2 150 4.90 5.15 D 2 100 6.50 6.40 E 3 80 25.00 24.60...
-
American Food Services, Incorporated leased a packaging machine from Barton and Barton Corporation. Barton and Barton completed construction of the machine on January 1, 2024. The lease agreement for...

Study smarter with the SolutionInn App


