When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain.
Question:
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain.
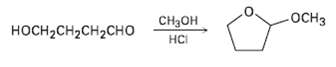
Transcribed Image Text:
-ОСHЗ Cнон носHCH2сH2CHо нCі
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
4Hydroxybutanal forms a cyclic hemiacetal when the hydroxyl ox...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
When benzene is treated with I 2 in the presence of C u Cl 2 , iodination of the ring is achieved with modest yields. It is believed that C u Cl 2 interacts with I 2 to generate I + , which is an...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce...
-
Analyze how you will use the challenge the process and enable others to act practices to improve the three leadership areas that you selected in module one. ? select one leadership theory or approach...
-
1. What sources of conflict were present in this scenario? 2. On PPT 9-4, The Conflict Process, overt behaviors are depicted as indicators of manifest conflict. What overt behaviors did you identify...
-
1. Gear Company records $2,000 of depreciation under the sum-of-years'-digits method in 2019, the compa- Cha ny's first year of operations. In 2020, the company decides to change to the straight-line...
-
Consider the plot of power as a function of effect size for the two-sample t-test shown in Figure 4.3. a. Create a plot to show power as a function of both effect size and sample size while keeping...
-
The rectangular loop of Wire shown in fig. bas a mass of 0.15 g per centimetre of length and is pivoted about side ab on a frictionless axis. The current in the wire is 8.2 A in the direction shown....
-
New Center Co . has concluded a contract with the Defense Department for $ 4 9 5 , 0 0 0 with a forecasted cost of $ 3 7 5 , 0 0 0 . It is estimated at this time that 6 0 % of the job is complete...
-
The Robbins Corporation is an oil wholesaler. The firms sales last year were $1 million, with the cost of goods sold equal to $600,000. The firm paid interest of $200,000 and its cash operating...
-
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example, (a) How would you prepare...
-
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are more reactive than ketones toward...
-
Determine all Taylor polynomials of f (x) = x 4 + x + 1 at x = 2.
-
A company reported the following: Net income 270,000 Preferred Dividends 10,000 Shares of common stock outstanding 20,000 Market price per share of common stock 36.40 Calculate the company's...
-
You have been asked to prepare a December cash budget for Ashton Company, a distributor of exercise equipment. The following information is available about the companys operations: The cash balance...
-
Green Company provides the following information about its single product. Targeted operating income Selling price per unit Variable cost per unit Total fixed cost $653,010 $7.45 $5.80 $98,010 How...
-
4. At the end of its first year of operations, December 31, 2025, Wildhorse Inc. reported the following information. Accounts receivable, net of allowance for doubtful accounts $867,500 Customer...
-
How do social institutions perpetuate and reproduce systems of inequality, and what mechanisms are at play in the institutionalization of privilege and marginalization?
-
In the context of Example 7.8, let \(\left\{W_{n}ight\}_{n=1}^{\infty}\) be a sequence of independent and identically distributed bivariate random vectors from a distribution \(F\) having mean vector...
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
The highest bond-dissociation energy is found in (a) O 2 ; (b) N 2 ; (c) Cl 2 ; (d) I 2 .
-
Write a structural formula for the product from each of the following reactions. (a) (b) (c) (d) (e) (f) NaOEt EtOH NaOEt EtOH NaOEt EtOH NaOEt EtOH (1)NaOEt, EtOH
-
Show all steps in the following syntheses. You may use any other needed reagents but you should begin with the compound given. (a) (b) (c) OEt OEt OEt OEt OEt
-
Provide the starting materials needed to synthesize each compound by acylation of an enolate. (a) (b) (c) CO2Et
-
Solve the triangle shown to the right. Round the lengths of sides to the nearest tenth and angles to the nearest degree. 96 b=5 a=7 B A C C9.0 (Do not round until the final answer. Then round to the...
-
Hello, need help with making the program MealData.txt Avocado Egg Rolls 6.00 Parmesan-crusted Chicken 19.75 Strawberry Shortcake Pizookie 8.25 BBQ Tri-Tip Sliders 12.95 Spicy Peanut Chicken with Soba...
-
Assume you have been appointed to develop ethnic and multicultural marketing for a small chain of household appliance stores in large metropolitan area. There are several large concentrations of...

Study smarter with the SolutionInn App


