Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c)
Question:
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds:
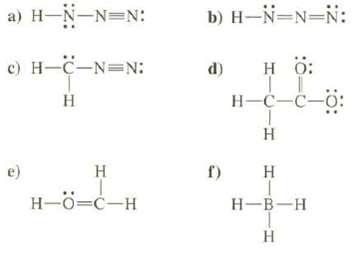
Transcribed Image Text:
a) H-N-N=N: c) H-C-N=N: H e) H H-Ö=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (19 reviews)
1 1 0 a HNNN 0 H ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species: a) H H-C-0_H d) H H C=N=N: b) H H-C-0: f) SF: : | H-NC-C- ...
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
A certain radioactive isotope is a by - product of some nuclear reactors. Due to an explosion, a nuclear reactor experiences a massive leak of this radioactive isotope. Fortunately, the isotope has a...
-
Explain the meaning of supply chain and provide an example to illustrate your answer. Your example should focus on a particular industry and involve at least one answer customer.
-
Consider a day when the Dow Jones industrial average went up 149.82 points. The following table shows the stock price changes for a sample of 12 companies on that day. a. Compute the sample variance...
-
I want sales representatives who can stand on their own. Once they have been through training and show how to apply their knowledge, it shouldnt be necessary for me to constantly tell them how they...
-
Outdoor Texas makes umbrellas, gazebos, and chaise lounges. The company uses a traditional overhead allocation scheme and assigns overhead to products at the rate of $ 30 per direct labor hour. The...
-
(30 Points) Consider the network in Figure-4 and Calculate the node-betweenness of node B and F. E F B Figure 4 C D
-
On April 1 Frank Knight incorporated and capitalized with $40,000 of his savings Waltham Oil and Lube Centers, Inc. On the same day, he signed a lease and operating agreement to operate the recently...
-
Show Lewis structures for these compounds: (a) CH5N (b) C 2 H 5 Cl (c) N 2 (d) CH 2 S (e) C 2 H 3 F (f) CH 4 S
-
Explain which of the two following structures would be more stable. Explain whether they represent isomers or are resonance structures. HIN: H N-H H :0: N-H HIN
-
(a) If X takes on the values 0, 1, 2, and 3 with probabilities 1/125, 12/125, 48/125 , and 64/125 , find E(X) and E(X2). (b) Use the results of part (a) to determine the value of E[(3X + 2)2].
-
An educator claims that less than 20% of college graduates smoke. Formulate the null and alternative hypotheses for a hypothesis test. State clearly the two possible conclusions that address the...
-
Newer processors such as Intel's i7 Kaby Lake include support for AVX2 vector/multimedia instructions. Write a dense matrix multiply function using single-precision values and compile it with...
-
People with higher resting pulse rates (beats per minute) tend to have higher IQ scores. For Exercises 912, determine whether the stated causal connection is valid. If the causal connection appears...
-
What is the approximate capacitance of the charge center + ground system? A. \(6 \times 10^{-8} \mathrm{~F}\) B. \(2 \times 10^{7} \mathrm{~F}\) C. \(4 \times 10^{6} \mathrm{~F}\) D. \(8 \times...
-
When the body absorbs nicotine, it converts it into cotinine. Experiments have ruled out coincidence as an explanation for a correlation between exposure to second-hand smoke and cotinine in the...
-
Amador Ltd is considering investing $\$ 120,000$ in equipment that has a life of 15 years. Its final scrap value is $\$ 25,000$. The equipment will be used to produce 15,000 deluxe pairs of rugby...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
Use the laws of logarithms to evaluate log 2 80 log 2 5.
-
Divalent carbon species called carbenes care capable of fleeting existence. For example, methylene: CH2, is the simplest carbene. The two unshared electrons in methylene can be either spin-paired in...
-
There are two different substances with the formula C4H10. Draw both, and tell how they differ.
-
There are two different substances with the formula C3H6. Draw both, and tell how they differ.
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


