Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the
Question:
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species:
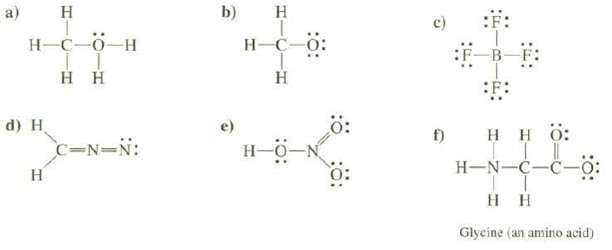
Transcribed Image Text:
a) H H-C-0_H Η Η d) H H C=N=N: b) H H-C-0: Η Ξ f) SF: Η Η Ο: | H-NC-C- Τ Η Η Glycine (an amino acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The number of valence electrons is the same as the group number of the atom Unshared electron...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
Montage Pty Limited (Montage) is a resident private company and is not a base rate entity. Which of the following transactions would result in a debit entry to Montage's franking account? Payment of...
-
Sweety Tea (ST) manufactures a range of designer T-shirts, which it produces to order for major department stores. On to March the sales department at ST received an order from retail company, Legend...
-
Do you dislike waiting in line? Supermarket chain Kroger has used computer simulation and information technology to reduce the average waiting time for customers at 2300 stores. Using a new system...
-
You are a small manufacturer of specialty medical devices located in Chicago, Illinois, and the market for your product is cardiologists in the United States. Does globalization affect your business?...
-
A car dealer presently leases a small computer with software for $5000 per year. As an alternative he could purchase the computer for $7000 and lease the software for $3500 per year. Any time he...
-
(A) We are given three diffent vectors: A = 6-3, B=-11+5j and C = A + B. (1) C = 2, (2) C = 51, (3) C = 5 + 2), (4) C=7+8 What is C in component form? A. 3 B. 1 C. 4 D. 2 Using the same three vectors...
-
Dixon Robotics manufactures three robot models: the A3B4, the BC11, and the C3PO. Dixon allocates manufacturing overhead to each model based on machine hours. A large portion of the companys...
-
Using Lewis structures show a balanced equation for the reaction of H2O with HCl.
-
Predict which of the following constitutional isomers for the compound that is formed from one atom each of hydrogen, oxygen, and chlorine is more stable: H-CI-: or H-O-CI:
-
Determine the magnitude of the hydrostatic force acting on gate AB, which has a width of 1.5 m. Water has a density of = 1 Mg/m. B -1.5 m- 2 m
-
In this chapter, personal selling is defined as (1) one-on-one, door-to-door engagement of key groups and stakeholders and (2) the ability to sell ones image and expertise. Discuss practical examples...
-
You fire a bullet of inertia \(m\) into the block shown in Figure P9.43. The bullet is initially traveling at speed \(v\), and the inertia of the block is \(4 \mathrm{~m}\). The surface on which the...
-
Cluppins and Raddle form a partnership on 1 November 2017, preparing accounts to 31 May each year. Bardell is admitted as a partner on 1 January 2019. Cluppins leaves the partnership on 29 February...
-
In the classic business movie Office Space, watch Scene 13, Flair. This communication does not go very well. Identify the communication barriers in this scene.
-
A 1.5 V D-cell battery is rated at \(15,000 \mathrm{~mA} \cdot \mathrm{h}\). For how long could such a battery power a flashlight bulb rated at \(1.5 \mathrm{~W}\) ?
-
Alameda Ltd has two divisions, Division Alpha and Division Beta. Division Alpha makes widgets, which it can sell externally for $\$ 45$ as well as internally to Division Beta. The marginal cost of...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Express ln a + 1/2 ln b as a single logarithm.
-
The following model is a representation of asparatame, C14H18N2O5, known commercially as NutraSweet. Only the connection between atoms is shown; multiple bonds are not indicated. Complete the...
-
How many valence electrons does the each of the following dietary trace elements have? (a) Zinc (b) Iodine (c) Silicon (d) Iron
-
Give the ground-state electron configuration for each of the following elements: (a) Potassium (b) Arsenic (c) Aluminum (d) Germanium
-
9. An arrow 2.5 cm high is placed at a distance of 25 cm from a diverging mirror of focal length 20 cm., Find the nature, position and size of the image formed. 10. The image formed by a convex...
-
1. Find the focal length of a convex mirror of radius of curvature 1m. 2. Focal length of a convex mirror is 50 cm. What is its radius of curvature? 3. Radius of curvature of a concave mirror is 25...
-
When an object is placed at the focus of a concave mirror, the image formed is (i) at infinity (ii) real and inverted, and (iii) highly magnified (or highly enlarged) M E A D C B P F At infinity N

Study smarter with the SolutionInn App


