Draw the cis and trans stereo isomers of this compound and explain their relative stabilities. OH CH
Question:
Draw the cis and trans stereo isomers of this compound and explain their relative stabilities.
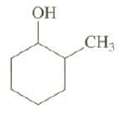
Transcribed Image Text:
OH CH₁
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The transstereoisomer is more stabl...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the structure for the cis and trans isomers of a. 1,3-dibromocyclopentane b. 1-chloro-2-methylcyclopropane
-
Draw the cis and trans isomers for the following compounds: a. 1-ethyl-3-methylcyclobutane b. 2-methyl-3-heptene c. 1-bromo-4-chlorocyclohexane d. 1,3-dibromocyclobutane
-
Draw the cis and trans isomers of the compound 3-methyl-3-hexene.
-
Find the dy/dx for the following 3 2x+1.2x+1 (x +1)4 1- y=-
-
Eye in the Sky Lens Company manufactures sophisticated lenses and mirrors used in large optical telescopes. The company is now preparing its annual profit plan. As part of its analysis of the...
-
Street, Rhode and Close carried on business in partnership sharing profits and losses, in the ratio 5 : 4 : 3. Their draft statement of financial position as on 31 March 20X2 was as follows: Street...
-
Why is dynamic modeling based on objects, but not classes?
-
Rivera, Sampson, and Elliott are partners in a commercial plumbing business. Rivera and Sampson have also started another contracting company and have cash flow needs which require periodic...
-
Suppose you borrowed $12,000 at a rate of 9.6% and must repay it in 5 equal installments at the end of each of the next 5 years. How much interest would you have to pay in the first year?
-
Determine the number of terms necessary to approximate cos x to 8 significant figure using the Maclaurin series approximation Calculate the approximation using a value of x = 0.3?. Write a program to...
-
Draw the chair conformations of 1, 1, 3trimethyl cyclohexane. Which conformation is more stable? Why is it not possible on the basis of the material in this chapter to determine the exact energy...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
What functional-, business-, and corporate-level strategies did GM adopt to regain its competitive advantage in the 1980-1990s? General Motors (GM) was once the largest and most profitable industrial...
-
Which of the following factors least affects a defined contribution plan participants account balance? A. Investment return. B. Inflation. C. Employer contributions. D. Life expectancy.
-
Short-term (one-year) interest rates over the next 3 years are expected to be 2%, 3%, and 3.55%. If you are ready to buy a three-year bond that yields 3%, what is your minimum required liquidity...
-
Refer to the previous exercise. What is the implicit required rate of return if dividends are expected to grow at a 5% annual rate? Previous exercise Calculate the stock price of OSE Water Co. if the...
-
Explain why you would be more or less willing to buy a share of Verizon stock in the following situations: a. Your wealth falls. b. You expect it (Verizon stock) to appreciate in value. c. The bond...
-
Susan Jones, owner of HyperTech, LLC decided that her storefront windows need to be washed in order to attract more foot traffic. The business sells iDevices, which are visible from the windows on...
-
Why did the world economy become more integrated? As you read this, is integration growing or retreating? What are the implications of these questions for international managers?
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
In Exercises 16, compute '(x) using the limit definition. (x) = x 2 + 3x
-
One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (1) Protonation of diazomethane by the carboxylic...
-
The hydrolysis of a biological thioester to the corresponding carboxylate is often more complex than the overall result might suggest. The conversion of succinyl CoA to succinate in the citric add...
-
One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehydes 3-phosphate. The process occurs by phosphorylation with...
-
Develop a SWOT analysis of Coles potential business. b. Can Cole be profitable and if so, in what period of time? c. What are the major business assumptions that require verification? d. Does Cole,...
-
The current temperature is 12 oC and the dew point is -29 oC. What is the relative humidity in percentage? Answer to one decimal place. The current temperature is 12 oC and the dew point is -29 oC....
-
The density of the block depends on what material the block is made out of. The table below shows the average densities for 3 different kinds of Earth materials. Using these data, calculate the...

Study smarter with the SolutionInn App


