Explain the difference in the percentages of the products in these two hydroborationreactions: CH3 CH, QH CH,
Question:
Explain the difference in the percentages of the products in these two hydroborationreactions:
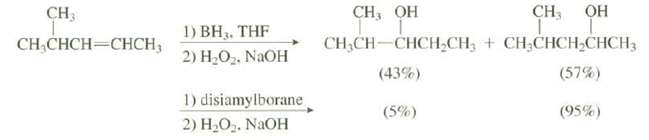
Transcribed Image Text:
CH3 CH, QH CH, ОН 1) BH3. THF 2) H,O,, NAOH CH,CH-CHCH-CH; + CH;CHCH,CHCH3 (43%) CH,CHCH=CHCH, (57%) 1) disiamylborane (5%) (95%) 2) H2O. NAOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Addition reactions involving boranes are regioselective and are greatly influenced by sterics Si...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain the difference in project time management and project scope management for projects using agile methods.
-
Explain the difference in intended application between strategic pricing and life-cycle costing.
-
In 75 words or fewer, explain the difference in calculating cost of goods sold using the FIFO, LIFO, and weighted- average inventory costing methods when using a perpetual inventory system.
-
The Kc for the following reaction is 9.30 X 10^-2 at 25C:PCl5(g) <-> PCl3(g) + Cl2(g) How many moles & grams of PCl5 must be added to a 2-literflask to obtain a Cl2 concentration of 0.150M...
-
What is intelligence gathering?
-
(a) Find the amount of heat that must be extracted from 1.3 kg of steam at 120 C to convert it to ice at 0.0 C. (b) What speed would this 1.3-kg block of ice have if its translational kinetic energy...
-
A stock price \(S\) is governed by where \(z\) is a standardized Wiener process. Find the process that governs dS as dt +bs dz.
-
Al Bundy is evaluating a new advertising program that could increase shoe sales. Possible outcomes and probabilities of the outcomes are shown next. Compute the coefficient ofvariation. Possible...
-
The December 31, 2024, unadjusted trial balance for the Wolkstein Drug Company is presented below. December 31 is the company's year-end reporting date. Account Title Cash Accounts receivable Prepaid...
-
How are network computers different from traditional personal computers? Describe some usage scenarios in which it is advantageous to use network computers.
-
An unknown compound has the formula C7H12. (a) What is the DU for this compound? (b) The unknown reacts with H2 in the presence of Pd to give C7H16. What information dos these provide about the...
-
Explain why this reaction occurs with anti-Markovnikov regiochemistry: CI + CF,CH,CH CF;CH=CH2 + HCI
-
When 0.030 mol HCl dissolves in 100.0 g benzene, the solution freezes at 4.04 C. When 0.030 mol HCl dissolves in 100.0 g water, the solution freezes at -1.07 C. Use the data in Table 12.4 to complete...
-
What will be the moment of inertia of a homogeneous triangular board of mass m with side lengths a and b when rotated about the axis passing through side a? mb mb mb mb (a) (b) 2mb 3 (c) (e) 3 12 2 6...
-
10. A ray of light falls on a horizontal plane mirror as shown. 40 In order to reflect the light ray vertically upwards, the plane mirror should be rotated by A. 20 B. 25 C. 40 D. 50
-
You are at a funhouse at Hershey Park. The ride you are on is a huge spinning platform.Suppose you move towards the outside edge of the spinning platform. What would happen to the angular momentum of...
-
A tennis ball is tossed straight up into the air. In addition to the force of gravity, the tennis ball is subject to a frictional force due to air friction. The time the tennis ball takes to reach...
-
The capacitor in the circuit is charged to 20 V at the time the switch is closed. (Figure 1) Figure 20 V C 1 0 12 x 10 i 20 1 of 1 80 www Part A If the capacitor ruptures when its terminal voltage...
-
In a column in the Wall Street Journal, two economists at the Council on Foreign Relations argue: Simply put, the Fed must choose between managing the level of reserves and managing rates. It cannot...
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
Consider the following reaction at a certain temperature: An equilibrium mixture contains 1.0 mole of Fe, 1.0 10 -3 mole of O 2 , and 2.0 moles of Fe 2 O 3 all in a 2.0-L container. Calculate the...
-
Simple sugars undergo reaction with phenyl hydrazine, PhNHNH2, to yield crystalline derivatives called osazones. The reaction is a hit complex however, as shown by the fact that glucose and fructose...
-
When heated to 100C, n-idose undergoes a reversible loss of water and exists primarily as 1, 6-anhydro-D-idopvranose. (a) Draw D-idose in its pyranose form, showing the more stable chair conformation...
-
Acetyl coenzyme A (acetyl CoA) is the key intermediate in food metabolism. What sugar is present in acetylCoA? NH2 N: CH N HsCH2CH2N,H2NHCHCH,2 - 0=P-0- Acetyl coenzyme A
-
Factory overhead of $44,700 consists of Indirect labor of $21,900, Depreciation expense-Factory of $16,900, and Factory utilities of $5,900. a. Compute total manufacturing costs. b. Prepare a...
-
In Giannakakos et al. (2016), titled Using Video Modeling with voiceover Instruction plus feedback to train staff to implement direct teaching procedures, why do you think collecting data on the...
-
Presidio, Inc., produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidios cost data table. 2. Calculate Presidios contribution margin ratio and...

Study smarter with the SolutionInn App


