Explain why this reaction occurs with anti-Markovnikov regiochemistry: CI + CF,CH,CH CF;CH=CH2 + HCI
Question:
Explain why this reaction occurs with anti-Markovnikov regiochemistry:
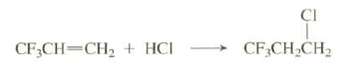
Transcribed Image Text:
CI + CF,CH,CH CF;CH=CH2 + HCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
In this reaction the primary carbocation intermediate is ...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why the reaction shown in eq. 7.19 occurs much more easily than the reaction (That is, why is it necessary to protonate the alcohol before ionization can occur?) (CH) C-OH(CHCHO
-
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole, but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene. piperidine
-
Explain why nitration of quinoline (eq. 13.7) occurs mainly at C5 and C8. No 2 (13.7 0C 5-nitroquinoline NO2 8-nitroquinoline
-
MULTIPLE CHOICE: 6. The stage of production at which the individual jointproducts are identified is referred to as the: A. Split-off point B. Joint point C. Separate identification point D. Relative...
-
What is BI governance?
-
A 2.50% grade meets a +2.50% grade at station 4 + 200 and elevation 293.585 m, 300-m curve, stakeout at 30-m increments. Tabulate station elevations for an equal-tangent parabolic curve for the data...
-
Casey Deesel is a sports agent negotiating a contract for Titus Johnston, an athlete in the National Football League (NFL). An important aspect of any NFL contract is the amount of guaranteed money...
-
The Outdoor Sports Company produces a wide variety of outdoor sports equipment. Its newest division, Golf Technology, manufactures and sells a single product: AccuDriver, a golf club that uses global...
-
Examine each of the following independent situations and state whether control does or does not exist. Where there is control, you are to state which party controls the entity. Give reasons for your...
-
A slab with a wet weight of 5 kg originally contains 50 percent moisture (wet basis). The slab is 600 by 900 by 75 mm thick. The equilibrium moisture content is 5 percent of the total weight when in...
-
Explain the difference in the percentages of the products in these two hydroborationreactions: CH3 CH, QH CH, 1) BH3. THF 2) H,O,, NAOH CH,CH-CHCH-CH; + CH;CHCH,CHCH3 (43%) CH,CHCH=CHCH, (57%) 1)...
-
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
-
Use the diagram to find the indicated measure. Find the area of OM. K 50 M A = 56.87 cm L
-
What are the best takeaways we can take from situational leadership. b) How can situational leadership help us in our workplace. c) How can we turn situational leadership into habits.
-
Jeff purchased a card for $180 that gives him 20 visits to a new gym and includes a one time fee for unlimited use of the sauna. After 5 visits, he has $123.75 left on the card, and after 11 visits,...
-
URSING PEER INTERVIEWS ASSIGNMENT INSTRUCTIONS OVERVIEW explore delegation policies as listed on the State Board of Nursing website for the state in which you hold your nursing license. You will also...
-
15. An empty swimming pool with a square floor dimension has a volume of 3y m. Given the weight of air in the swimming pool is about 500 N and the air density is 1.29 kg/m. (a) (b) Find the length of...
-
Christensen spoke in depth about how disruptive technologies can affect existing market leading companies. Considering this, explain how 3D Printing has created a new market and how an existing...
-
Figure18 . 25 shows the series of Walmart daily closing prices between February 2001 and February 2002 (thanks to Chris Albright for suggesting the use of these data, which are publicly available,...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Write the equilibrium expression (K) for each of the following gas-phase reactions. a. N(g) + O(g)=2NO(g) b. NO(g)2NO(g) c. SiH4(g) + 2Cl(g) d. 2PBr3(g) + 3Cl(g) SiCl4(g) + 2H(g) 2PC13(g) + 3Br(g)
-
One of the steps in the biological pathway for carbohydrate metabolism is the conversion of fructose 1, 6-bisphosphatc into dihydroxyacetone phosphate and glyceraldehydes 3-phosphate. Propose a...
-
L-Fucose, one of the eight essential monosaccharide?s, is biosynthesized from GDP-D-mannose by the following three-step reaction sequence, where (H)P?guano sine diphosphate (a ribonucleoside...
-
Of the 19 L amino acids, 18 have the S configuration at the carbon. Cysteine is the only L amino acid that has an R configuration. Explain.
-
A project costs $80 MM, with 25% of the investment int in year 1 and the balance in year 2. The plant operates atates at 50% capacity in year 3 and then full capacity for the next next 20 years. The...
-
What is organizational Behaviour the study of? How is social sciences related to organizational behavior? What is the difference between social science and organizational behavior? What is the...
-
Describe the nature of organization change, including forces for change and planned versus reactive change

Study smarter with the SolutionInn App


