Explain which compound you expect to have the higher melting point. c) of b) CH(CH)1CH or or
Question:
Explain which compound you expect to have the higher melting point.
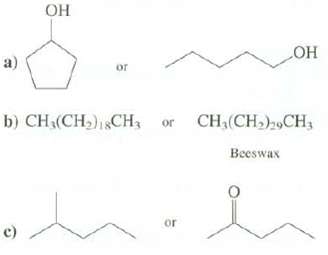
Transcribed Image Text:
ОН c) of b) CH₂(CH₂)1CH₁ or or ОН CH₂(CH₂)29CH, Beeswax
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
a The cyclic alcohol mp 19C has a higher melting point than the straight chain ...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which compound you expect to have the higher boiling point. a) cont b) CH3CHCOH c) CH,CH,OCH,CH, d) CH H or or CHCHCHCH or CHCH,CH,OCH,CH,CH, or H
-
Which compound would you expect to have the higher melting point, propane or cyclopropane? Explain your answer.
-
Explain which compound has the higher melting point or boiling point: a) Melting point b) Boiling point or C COCH c) Boiling point or
-
Shemekia applied to a college that uses multiple regression to select students. This college only considers students whose predicted firstyear GPA is 3.0 or higher. Shemekias predicted GPA was 2.9...
-
Grant Lawson has just been appointed as the new financial controller of Safety Chemicals Ltd, which has three separate divisions Industrial Chemicals, Paints, and Household Chemicals. During his...
-
Use a computer to find the following probabilities. a. P(t64 > 2.12) b. P(t27 > 1.90) c. P(t159 > 1.33) d. P(t550 > 1.85)
-
Explain what the expected frequencies represent in a goodness-of-fit test.
-
On January 2, 2008, two identical companies, Daggar Corp. and Bayshore Company, lease similar assets with the following characteristics: 1. The economic life is eight years 2. The term of the lease...
-
Ty has been working with toddlers for 2 years after receiving a CDA from the national organization, Council for Professional Recognition. Ty's reviews reveal a basic understanding of child...
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
Explain the difference in the melting points of these isomers: mp=-140C 0 mp = 7C
-
Which of these two salts would your expect to be more soluble in hexane(C6H14)? NH, CI + Or N(CHCHCHCH3)4 Cl
-
Imagine you and your teammates are buyers for a firm such as Applebees, Dicks Sporting Goods, Hilton Hotels, or another company you like. Map out a logical buying process for a newtask purchase for...
-
A 7.0 C charge is moving to the right at a speed of 49m/s. A B-field is then induced with a strength of 3.7 T directed out the page. What is the magnitude of the force exerted on the wire? What...
-
3. Calculate the density of (aluminum, brass, lead or any other materials) given to you by your instructor using Eq.1 and its uncertainty Ap. maV+Vam Ap- 4. Within how many standard deviations does...
-
The following items are reported on a company's balance sheet: Cash $296,600 Marketable securities 185,000 Accounts receivable (net) 121,000 Inventory 127,000 Accounts payable 228,000 Determine (a)...
-
Suppose you write a June expiration call option on Tesla with exercise price $85 and at the same time, write a June Tesla put option with exercise price $80. The premium of the call option is $0.90...
-
Nakashima Gallery had the following petty cash transactions in February of the current year. Nakashima uses the perpetual system to account for merchandise inventory. February 2 Wrote a $340 check to...
-
One method for growing thin silicon sheets for photovoltaic solar panels is to pass two thin strings of high melting temperature material upward through a bath of molten silicon. The silicon...
-
Citing a scientific article, explain in your own words, how DNA fingerprinting has been used in forensic science to solve crimes and why it may not always be accurate or effective.
-
For the region S in Example 1, show that the sum of the areas of the upper approximating rectangles approaches 1/3, that is, Data from Example 1 Use rectangles to estimate the area under the parabola...
-
Methyl aryl ethers, such as anisole, are cleaved to iodomethane and a phenoxide ion by treatment with LiI in hot DMF. Propose a mechanism for this reaction.
-
Tert-Butyl ethers can be prepared by the reaction of an alcohol with 2-methyipropene in the presence of an acid catalyst. Propose a mechanism for this reaction.
-
Meerwein?s reagent, triethyloxonium tetra-fluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein?s reagent with...
-
Discuss the innovative idea/project addressing the problem below by the following (a) potential impact - (b) novelty and innovation- (c) feasibility- (d) sustainability- Reference: i. Potential...
-
Marianne's duplex sold at a foreclosure auction for $280,000. At the time of foreclosure, she had not made any payments to the bank for 11 months ($2,400 each). The outstanding balance of her...
-
Find the complete solution of the linear system, or show that it is inconsistent. (If the system has infinitely many solutions, express your answer in terms of t, where x = x(t), y = y(t), and z=t....

Study smarter with the SolutionInn App


