Which of these two salts would your expect to be more soluble in hexane(C6H14)? NH, CI +
Question:
Which of these two salts would your expect to be more soluble in hexane(C6H14)?
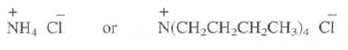
Transcribed Image Text:
NH, CI + Or N(CH₂CH₂CH₂CH3)4 Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The salt with the nonpolar hydr...View the full answer

Answered By

Palash Ghorai
Not much experience but try to solve every problem.Try my best
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which alkyl halide would you expect to be more reactive in an SN2 reaction with a given nucleophile? In each case, you can assume that both alkyl halides have the same stability a. CH3CH2CH2Br or...
-
Which alkyl halide would you expect to be more reactive in an E2 reaction? a. b. c. d. CH3 CHCHCHCH.CH, or CH.CHCH CHCH. Br Br Br CH3CH2CH CHCH3 or CH3CH2CHCH CH3 CH2CHCH2CH3 or CH2CH CHCH3
-
Which alkyl halide would you expect to be more reactive in an SNI solvolysis reaction? CH2CH3 CH3 or - CH CHCH H CH CH2CH H
-
You are asked to evaluate the following two projects for Boring Corporation using the NPV method combined with the PI approach, which project would you select? Use a discount rate of 10 percent....
-
The onstt.t0 Construction Company is a real estate developer and building contractor. The company has two sources of long-term capital: debt and equity. The cost of issuing debt is the after-tax cost...
-
Southwestern University (SWU), a large state college in Stephenville, Texas, enrolls close to 20,000 students. The school is a dominant force in the small city, with more students during fall and...
-
Explain the difference between the Address and Addresses classes.
-
Tidy House produces a variety of household products. The firm operates 24 hours per day with three daily work shifts. The first-shift workers receive regular pay. The second shift receives an 8...
-
This is a research to investigate the FAA maintainability requirements for aviation systems. Begin by considering the following statement from the United States General Accounting Office Testimony,...
-
Dynamic Mattress decides to lease its new mattress stuffing machines rather than buy them. As a result, capital expenditure in the first quarter is reduced by $50 million, but the company must make...
-
Explain which compound you expect to have the higher boiling point. a) cont b) CH3CHCOH c) CH,CH,OCH,CH, d) CH H or or CHCHCHCH or CHCH,CH,OCH,CH,CH, or H
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
What is a trust? Name five possible advantages of using trusts in estate planning.
-
A steam turbine operates on a superheated Rankine cycle. The pressure and temperature of the steam leaving the boiler are 10 bar and \(350^{\circ} \mathrm{C}\) respectively. The specific steam...
-
The condenser pressure of the turbine in P3.2 is reduced to 0.15 bar. Calculate the same parameters for this cycle as in the previous example. Why have the parameters improved so much? [28.97\%;...
-
Compute the mean FEV1 for those with asthma and those without. What is counterintuitive about the result?
-
depicts a closed cycle gas turbine operating on the Joule cycle (i.e. constant pressure heat addition and rejection, and isentropic compression and expansion). Energy is added to the working fluid...
-
An amount of pure substance equal to \(1 \mathrm{kmol}\) undergoes an irreversible cycle. Neglecting the effects of electricity, magnetism and gravity, state whether each of the following...
-
The stock market of the late 1990 s and early 2000 s represented a period of irrational exuberance. Investors invested as they never had, egged on by analysts who could say no evil of the companies...
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
If f is the function whose graph is shown in Figure 2 and g(x) = 0 x f(t) dt, find the values of g(0), g(1), g(2), g(3), g(4), and g(5). Then sketch a rough graph of g. Figure 2 y -2. 1 0 1 2 y =...
-
Acid-catalyzed hydrolysis of a 1, 2-epoxycyclohexane produces a trans-diaxial 1, 2diol. What product would you expect to obtain from acidic hydrolysis of cis-3-tert-butyl-1, 2-epoxycyclohexane?...
-
Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason...
-
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the same conditions yields Cyclohexanone. Propose mechanisms for both reactions,...
-
How do you prioritize features and attributes when designing a new product or service? What steps should be taken to ensure that the design process aligns with the organization's overall strategy and...
-
Information that does not have an adjustment reason code to route payments into the correct bank accounts to relay about patient benefit coverage to indicate the amount being paid and the date of...
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...

Study smarter with the SolutionInn App


