Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20
Question:
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene:
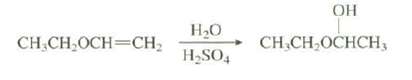
Transcribed Image Text:
OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (22 reviews)
The carbocation intermediate formed from this vinyl eth...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The alkene 3,3-dimethyl-1 -butene undergoes acid-catalyzed hydration with rearrangement. Use the mechanism of hydration and rearrangement to predict the structure of the hydration product of this...
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
Why does a heavy parachutist fall faster than a lighter parachutist who wears a parachute of the same size?
-
The membrane filter technique is used to test a polluted water sample for coliform group. Three different dilutions of the water sample were prepared and each was filtered through 5 filter membranes....
-
Discuss the major issues in implementing BI. Discuss.
-
An 90-m curve, g1 = 1.50%, g2 = +0.75%, VPI station = 6 + 280, VPI elevation = 550.600 m, stakeout at 10-m increments. Compute and tabulate full-station elevations for an unequal-tangent vertical...
-
Which of the following statements is true? a. Lasso regularization and ridge regularization are different methods for standardizing variables. b. Lasso regularization may prevent overfitting of a...
-
Marketers often target consumers before, during, or after a trigger event, an event in ones life that triggers change. For example, after having a child, new parents have an increased need for baby...
-
INFORMATION As a management accountant, you have recently been recruited to be part of the strategic management accounting team of GHK Accountants. GHK Accountants has been contracted to help...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Explain why this reaction occurs with anti-Markovnikov regiochemistry: CI + CF,CH,CH CF;CH=CH2 + HCI
-
The addition of HCl to alkynes proceeds through a vinyl cation intermediate. Explain which of the two possible vinyl cations that could be formed from the addition of HCl to propyne is morestable....
-
What was the total cost of Tootsie Rolls treasury stock at December 31, 2007? What was the amount of the 2007 cash dividend? What was the total charge to Retained Earnings for the 2007 stock dividend?
-
5. Arslan and Belgin consume two goods, q1 and 92. function is UA = 9A19A2 UB = 9B19B2 Their endowments are 9A = 10, 9A2 = 20, 9B = 20, 9B2 = 10. (a) Find the marginal rates of substation for each...
-
Compare Maslow's Hierarchy of Needs and Herzberg's Two-Factor Theory of motivation. How can these theories be applied to enhance employee motivation in the workplace?
-
What do you think is the most important aspect of international risk management for a manager? Why?
-
From your point of view and knowledge, what do you think about globalization, is it a benefit to the greater population of the world?
-
What are the differences and similarities in contingency leadership and situational leadership. How are contingency leadership and situational leadership important to how successful an organization...
-
Shipments of Household Appliances. The time plot in Figure 18 . 31 shows the series of quarterly shipments (in million dollars) of US household appliances between 1985 and 1989 (dataare available in...
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
For a typical equilibrium problem, the value of K and the initial reaction conditions are given for a specific reaction, and you are asked to calculate the equilibrium concentrations. Many of these...
-
The amino acid threonine, (2S, 3R)-2-amino-3-hydroxybutanoic acid, has two chirality centers. (a) Draw a Fischer projection of threonine. (b) Draw a Fischer projection of a threonine diastereomer,...
-
Hemoglobin has pI = 6.8. Does hemoglobin have a net negative charge or net positive charge at pH = 5.3? At pH = 7.3?
-
Show how you could prepare the following -amino acids from the appropriate carboxylic acids: (a) Phenylalanine (b) Valine
-
Explain organizational change and briefly discuss the three types of change? ( 350 words please)
-
Identify an organization that has experienced change Classify the type of organizational change the organization experienced Describe how the organization overcame the resistance to the change...
-
Carlton Bank has an increase in reserves of $1,000,000. If the reserve ratio is 10%, by what amount may Carlton increase its demand deposits?

Study smarter with the SolutionInn App


