For the assembly of Prob. 18.73, determine the dynamic reactions at A and B at t =
Question:
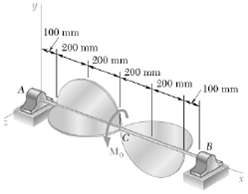
For the assembly of Prob. 18.73, determine the dynamic reactions at A and B at t = 2s.
Transcribed Image Text:
100 mm 200 mm 200 mm 200 mm 200 mm 100 mm B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Fixed axis rotation with constant angular acceleration a ai cx t 12aa 0 0 wi x282027 22 31416 ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Vector Mechanics for Engineers Statics and Dynamics
ISBN: 978-0073212227
8th Edition
Authors: Ferdinand Beer, E. Russell Johnston, Jr., Elliot Eisenberg, William Clausen, David Mazurek, Phillip Cornwell
Question Posted:
Students also viewed these Mechanical Engineering questions
-
Determine the normal reactions at A and B in Prob. 51.
-
Determine the normal reactions at A and B and the force in link CD acting on the member in Prob. 58.
-
Determine the components of the reactions at A and E when a 24- lb force directed vertically downward is applied (a) At B, (b) At D 2 in. 6 in. 2 in. R. 3 in. D. E. I in. 2 in.
-
Calculate the following ratios from the data given below: 1. Debt ratio 2. Debt service coverage multiples 3. Interest coverage Balance Sheet 12/31/2014 Assets Cash..$575,000 Short-term...
-
Jill Pederson is paid $800 for a 40-hour week and time-and-a-half for hours that exceed 40. Compute the employer's total expense of employing Jill for the 44 hours that she worked during the second...
-
A solution is 0.200 M in Co2+ and 0.0650 M in Cd2+. Calculate (a) The Co2+ concentration in the solution as the first cadmium starts to deposit. (b) The cathode potential needed to lower the Co2+...
-
Take the product of the perturbation velocity equation given by Eq. (17.53) by any component of the perturbation velocity. This results in an equation for \(v_{x}^{\prime} v_{y}^{\prime}\), which is...
-
Jane Myers is the manager of an extremely successful gift shop, Janes Gifts, which is operated for the benefit of local charities. From the data below, she wants a cash budget showing expected cash...
-
Why is Microsoft Corp bond a good investment? The Bond has a maturity date of 6/1/2039 and a coupon rate of 5.2000% and is At the current price of 114.234 USD, this equates to a 4.16 percent annual...
-
The following information relates to Leonard, a middle-management accountant, not engaged in negotiating contracts, of a public corporation, Peter Productions Ltd. which is located in Ontario. (A)...
-
The assembly shown has a mass of 6 kg and consists of 4 thin 400-mmdiameter semicircular aluminum plates welded to a light 1-m-long shaft AB. The assembly is at rest ( = 0) at time t = 0 when a...
-
The assembly of Prob. 18.63 is initially at rest ( = 0) when a couple M0 is applied to axle AB. Knowing that the resulting angular acceleration of the assembly is = (150 rad/s2)i, determine (a) The...
-
Use the part of the scorecard that shows the distance in meters for the first four holes of the Millbrook Resort Golf Course in Queenstown, New Zealand. Determine the distances indicated. Hole 3,...
-
Business intelligence software supports decision making by presenting visual, interactive views of the state of a business. True or False
-
In what stage in the product life cycle would you consider Tesla automobiles to be? a. Introduction b. Growth c. Maturity d. Decline
-
Siobhan Clark just bought a Chicago Cubs jersey with player Jon Lesters name and number on the back, as well as a hat and jacket that display the Cubs logo. The type of branding strategy used for...
-
Sally and Brian only go shopping on Tuesdays because they can get the senior discount. Which pricing strategy are they taking advantage of? a. Predatory pricing b. Price discrimination c. Target...
-
How can consumers know if its safe to use their credit cards to make a purchase?
-
Rayes approach to rebalancing global equities is consistent with: A. the Laws being risk averse. B. global equities having higher transaction costs than other asset classes. C. global equities having...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
When treated with dilute HCl(aq), the solid that reacts to produce a gas is (a) BaSO 3 ; (b) ZnO; (c) NaBr; (d) Na 2 SO 4 .
-
A process requires a mixture that is 21 percent oxygen, 78 percent nitrogen, and 1 percent argon by volume. All three gases are supplied from separate tanks to an adiabatic, constant-pressure mixing...
-
A mixture that is 15 percent carbon dioxide, 5 percent carbon monoxide, 10 percent oxygen, and 70 percent nitrogen by volume undergoes an adiabatic compression process having a compression ratio of...
-
Propane and air are supplied to an internal combustion engine such that the air-fuel ratio is 16:1 when the pressure is 95 kPa and the temperature is 30C. The compression ratio of the engine is...
-
In this problem, you are asked to go through the steps of a finite element analysis. The key features include the use of isoparametric element formulation and Gauss numerical integration. (A) Problem...
-
A wheel rolls without slipping on a surface with a constant radius of curvature R. The centre of curvature is located at point O. R 20 9 G m Assume the wheel can be approximated as a uniform disk...
-
In this problem, use uniform modal damping (i with values to be determined as part of the problem. The other parameters are: mass of the base 95 kg, mass of the elbow joint and actuator m2 30 kg and...

Study smarter with the SolutionInn App


