Give the principal organic product formed in each of the following reactions. (a) Same as part (a)
Question:
Give the principal organic product formed in each of the following reactions.
(a) Same as part (a) with no poision
(b)
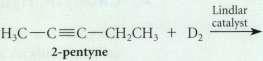
Transcribed Image Text:
Lindlar catalyst 2-pentyne
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a The product is the alkane ...View the full answer

Answered By

Swati gupta
Professionally I am going to become a Cost and Management Accountant very soon. I am a fresher in the area of tutoring but I am always admired and praised by my friends for teaching them. They always ask me to start giving tutoring services professionally. With the help of this platform, I am looking forward to give my best.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Write a structural formula for the principal organic product formed in the reaction of methyl bromide with each of the following compounds: (a) KOCH2CH3 (potassium ethoxide) (b) (c) LiN3 (lithium...
-
Write the structure of the principal organic product formed in the reaction of 1-propanol with each of the following reagents: (a) Sulfuric acid (catalytic amount), heat at 140C (b) Sulfuric acid...
-
Give the structure of the principal organic product formed by free-radical bromination of each of the following: (a) Methylcyclopentane (c) 2, 2, 4-Trimethylpentane (b)...
-
A product has a contribution margin of $6 per unit and a selling price of $30 per unit. Fixed costs are $24,000. Assuming the new technology increases the unit contribution margin by 60 percent but...
-
Milo Co. had 800,000 shares of common stock outstanding on January 1, issued 126,000 shares on May 1, purchased 63,000 shares of treasury stock on September 1, and issued 54,000 shares on November 1....
-
Shown in the figure is a gear-driven squeeze roll that mates with an idler roll, below. The roll is designed to exert a normal force of 30 lbf/in of roll length and a pull of 24 lbf/in on the...
-
For the adiabatic flash operation shown in Figure 10.34, calculate the following: (a) Change in availability function \(\left(T_{0}=100^{\circ} \mathrm{F} ight)\) (b) (b) Lost work (c) Thermodynamic...
-
High-low method and regression analysis. Happy Business College has recently opened a restaurant as part of its hospitality major. For the first 10 weeks the manager did not estimate any costs, but...
-
Some managers argue that international joint ventures/alliances are more costly than beneficial in terms of problems, making these ventures almost doomed to failure before beginning. Do you agree...
-
Dairy Services was organized on August 1, 20Y2. A summary of the revenue and expense transactions for August follows: Fees earned ..................................... $627,600 Wages expense...
-
Compare the results of hydroboration-oxidation and mercuric ion-catalyzed hydratioa for 2-butyne.
-
What product is obtained in each case when 3-hexyne is treated in each of the following ways? With H2 over Pd/C and quinoline and the product of that reaction with D2 over Pd/C
-
Describe three types of manipulations that could produce materially misleading financial statements.
-
A binary option (also called digital option) is an exotic option for which the payoff at maturity to the holder of the option is either some US dollar amount or nothing at all. One example of a...
-
Given the following factored polynomial, p(x)=x(2-x)(x+2)5 Perform the Multiplicity Test by answering the following: The zero x= -2 has multiplicity A/ The zero x= 0 has multiplicity The zero x= 2...
-
Water of 500C is cooled down to 20C. The process takes place isobarically at 5MPa. Determine the thermodynamic mean temperature of the heat source. Give a formula for a diffuser that describes the...
-
A roller coaster moves in different curves as it goes through the bumps in its track. We can easily analyze the forces at any point in the ride, by drawing a circle that matches up with the track at...
-
The reaction between HCI and NaOH is represented below. When equal volumes of 1.00 M NaOH and 1.00 M HCI are mixed, 57.1 kJ of heat is released. What would AH be if 2.00 M HCI and 2.00 M NaOH is used...
-
Refer to the data on garbage in Exercise 3.31. (a) Replace the year by (year - 1970). Calculate the correlation coefficient between (year - 1970) and amount of garbage in millions of tons. (b) Based...
-
Calculate Total Contribution Margin for the same items. Total Revenue Total Variable Costs Total Contribution Margin $50.00 a. $116.00 $329.70 b. $275.00 $14,796.00 $7,440.00 c. $40,931.25 d....
-
Draw a curved-arrow mechanism for each of the reactions given in Fig. P27.77. (a) (b) (c) (d) CH=O CH=0 o-phthalaldehyde CH 0 I CHCl + NH3 + Na+ SH + acetone H + HN-CH-CO2 + HSCHCHOH CH3 Figure...
-
Identify each of the compounds AD in the reaction scheme shown in Fig. P27.76. Explain your answers. i || || H3C-C-NH-CH-C-OEt I CH(CH3)2 Figure P27.76 HNNH, B A HC1, HO heat NaNO/HCI C B HO, "OH...
-
(a) For many years it was difficult to determine the X-ray structures of proteins that are imbedded in membranes because, when they are extracted into an aqueous buffer, they denature. Explain why...
-
Calendar Plc have a year end of 30/09/X2. They have prepared the following information about their non-current assets: Land was revalued to 4.2 million on 28/09/X2 All buildings are depreciated using...
-
Antique Accents reported the following on its 2020 income statement. Using the income statement and the balance sheet given below, prepare the statement of cash flows using the indirect method. For...
-
Jamal, Maggie, and Kendra are dividing an estate consisting of a house, a vacation home, and a small business. Their valuations are shown below. Determine the payment that Kendra receives from the...

Study smarter with the SolutionInn App


