Question: How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuricacid? H2SO4 CH3CH2CH2CCH2CH3 CH 3-Methyl-3-hexanol
How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfuricacid?
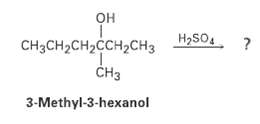
H2SO4 CH3CH2CH2CCH2CH3 CH 3-Methyl-3-hexanol
Step by Step Solution
3.44 Rating (151 Votes )
There are 3 Steps involved in it
HSO4 OH CH3CHCHCHCH HO CH3 CH3CH CHCH3 CC CH3 Z3Methyl3hexene ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-OC-A (138).docx
120 KBs Word File


