Question: How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are more reactive than ketones toward nucleophilicaddition)
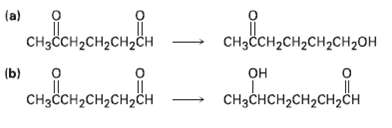
" (a) CHCH-CH2CH2CH2H CHH2H2CH2 (b) "one CCH H2H2 CCH-CH2CH,H
Step by Step Solution
3.51 Rating (154 Votes )
There are 3 Steps involved in it
In general ketones are less reactive than aldehydes for both st... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-AN (86).docx
120 KBs Word File


