Identify each of the following groups as an activator or deactivator and as and o,p-director orm-director. (e)
Question:
Identify each of the following groups as an activator or deactivator and as and o,p-director orm-director.
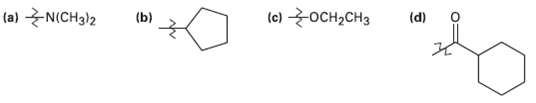
Transcribed Image Text:
(e) OCH2CH3 (a) N(CH3)2 (b) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Group a NCH32 b c CHCH3 d 0 Identification opactivator opactivator opactivator mdeactivat...View the full answer

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify each of the following groups as a population or a sample. If the group is a sample, identify the population to which the sample is related. A. The S&P MidCap 400 Index viewed as representing...
-
Identify each of the following as describing corporations, proprietorships, and/or partnerships. Some items have more than one answer. a. Distinct legal entity separate from its owners. b. More than...
-
Identify each of the following alkyl groups as being primary, secondary, or tertiary, and give it a systematic IUPAC name. (a) (b) (c) (d) (e) (f) CH CH3 CH3 CH CH2 CH2 CH3 CH3 CH3 CHCH CH3 CH2 CH3...
-
Use the data given in Table 3.9 to compute the mean, standard deviation, coefficient of variation, and coefficient of skewness for the current ratio of JNJ. Table 3.9 Year 1990 1991 1992 1993 1994...
-
William is employed by an accounting firm and uses his automobile in connection with his work. During the month of October 2016, he works at the office for 3 days and participates in the audit of a...
-
An open tubular column is 30.1 m long and has an inner diameter of 0.530 mm. It is coated on the inside wall with a layer of stationary phase that is 3.1 m thick. Unretained solute passes through in...
-
P(x < 180) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
Casebolt Company wrote off the following accounts receivable as uncollectible for the first year of its operations ending December 31, 2014: Customer Amount Shawn Brooke.......$ 4,650 Eve Denton...
-
Some time ago, you entered a forward contract to purchase 10 shares of Advanced Micro Devices, Inc. (AMD) for $32 per share. Your forward contract ends in 6 months and the current spot price of AMD...
-
The fin shown in Figure P3-56 has a base maintained at 300ºC and is exposed to the convection environment indicated. Calculate the steady-state temperatures of the nodes shown and the heat loss...
-
The following compound can?t be synthesized using the methods discussed in this chapter. Why not?
-
Predict the major product(s) of nitration of the following substances. Which react faster than benzene and which slower? (a) Bromobenzene (b) Benzonitrile (c) Benzoic acid (d) Nitrobenzene (e)...
-
Economists say that individuals make decisions at the margin. What does this mean?
-
What are some issues that could come up during the research and development stage of a new product that would be a reason to delay it's launch or give up on the idea completely?
-
Match the following terms to the correct definitions. Drag and drop application. Fraudulent financial reporting Fraudulent financial reporting drop zone empty. Misappropriation of assets...
-
Find the number of subsets of set S. S = {1, 2, 3, ..., 9}
-
The sale of land that was held for business-use purposes results in what type of gain or loss?
-
What are the Compliance-Based Codes of Ethics and Value-Based Codes of Ethics? Why are they important in International Ventures? Describe why responsible sourcing is included in many organizational...
-
Using a sector attribution analysis based on Exhibit 1, which US large-cap sector is the primary contributor to the portfolios underperformance relative to the benchmark? A. Utilities B. Consumer...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Redo Problem 5.29 using Aspen Plus. Problem 5.29 The inlet to an adiabatic compressor is nitrogen at 1 bar (0.1 MPa) and 150 K, and the exit pressure is 10 bar (1 MPa). a. Determine the minimum work...
-
Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
Suggest reagents that could be used to prepare these alkyl halides from alcohols: CH3 CH, b) CH,CI ) C,r Ph CH;CH2 Br CI Br ) d)
-
World Tourism Day 2 0 1 7 , with its official celebration on September 2 7 in Qatar, was focused on sustainable tourism. Established by the United Nations World Tourism Organization ( UNWTO ) , World...
-
Tracy is single and had adjusted gross income of $35,800 in 2018. Tracy also has the following items: How much may Tracy claim as itemized deductions?
-
Calculate the geometric mean return of an investment with five year returns of 10%, (15%), 12%, 8% and (5%).

Study smarter with the SolutionInn App


