Limonene, a major component of lemon oil, has the formula C10H16. (a) On reaction with excess H2
Question:
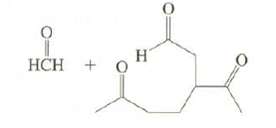
Limonene, a major component of lemon oil, has the formula C10H16.
(a) On reaction with excess H2 in the presence of Pt, limonene produces C10H20. What information does this provide about the structure of limonene?
(b) On ozonolysis, limonene produces these compounds. Suggest possible structures forlimonene.
Transcribed Image Text:
Н НСН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a The DU of limonene is 3 The DU of the hydrogena...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A major component of financial planning is to forecast future financial statements. If you had a company's balance sheets and income statements for the past 5 years but no other information, how...
-
A major component of gasoline is octane (c8h18). When liquid octane is burned in air it reacts with oxygen (o2) gas to produce carbon dioxide gas and water vapor. Calculate the moles of oxygen needed...
-
1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids that differ from each other by a single methyl or methylene group.? 3-Classify the 20 standard amino acids...
-
According to the American Red Cross, 11.6% of all Connecticut residents have Type B blood. A random sample of 28 Connecticut residents is taken. X = the number of Connecticut residents that have Type...
-
Grania knows that the pH of blood is normally 7.35 to 7.45. She sees that her blood test results show 7.57. Is Granias blood too acid or too alkaline or normal?
-
An electron and a proton are released from rest in space, far from any other objects. The particles move toward each other, due to their mutual electrical attraction. (a) When they meet, is the...
-
A stock price \(S\) is governed by the model where the period length is 1 month. Let \(u=\mathrm{E}[w(k)]\) and \(\sigma^{2}=\operatorname{var}[w(k)]\) for all \(k\). Now suppose the basic period...
-
The Whistle Stop Cafe in Weems, Georgia, is well known for its popular homemade ice cream, made in a small plant in back of the cafe. People drive all the way from Atlanta and Macon to buy the ice...
-
What is the output of the following code ? def f(n): return [[int(j=i) for j in range(n)] for i in range(n)] def show(mat): n=len(mat) for i in range(n): for j in range(n): print(mat[i][j],end=' ')....
-
what are the features detected by modernizr? CSS Reflections i. ii. iii. Web Workers SNIL iv. IndexedDB a. i and ii b. i, ii and iii c. i, ii and iv d. ii, iii and iv
-
Suggest a mechanism for thisreaction: CH,Br Br2 CH2=CHCH CH,CH,OH H,O
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
What information is contained in the specification K p = 10;000?
-
How do executive-level decisions affect organizational change? How should an executive cast vision for a major change? For example, when an executive wants to execute change in the organization, what...
-
Saleperson's client for lease, so sale person represent to a ) tenant b ) broker c ) lender d ) attoney
-
If USD depreciates by 5.88% against the Russian Ruble, then by how much has the Ruble appreciated against the USD? a. 6.52% O b. 6.25% O c. 5.88% O d. 5.50%
-
What should be the first step in managing third-party/supply chain risks? a. None of the above b. Establish processes for managing supply chain risks and assess their workability c. Communicate...
-
Which of the following statements is true? a. Futures contracts are less liquid than forward contracts, ceteris paribus. b. Futures contracts are not traded on an exchange. c. Currency options are...
-
What are the three categories of discount loans that the Fed gives to banks?
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
A popular chemical demonstration is the magic genie procedure, in which hydrogen peroxide decomposes to water and oxygen gas with the aid of a catalyst. The activation energy of this (uncatalyzed)...
-
The octapeptide angiotensin II has the sequence Asp-Arg-Val-Tyr-IIe-His-Pro-Phe. What fragments would result if angiotensin II were cleaved with trypsin with chymotrypsin?
-
What is the N-terminal residue on a peptide that gives the following PTH derivative on Edmandegradation?
-
Draw the structure of the PTH derivative that would he formed on Edman degradation of angiotensin II (Problem 26.12).
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


