Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,,
Question:
Show the products of thesereactions:
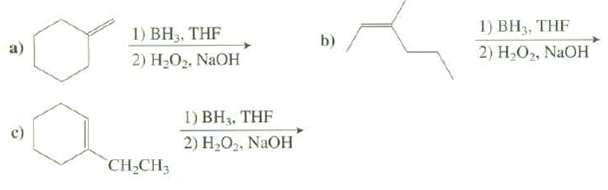
Transcribed Image Text:
1) BH3, THF 1) BH3. THF 2) Н.О. NaOH b) 2) Н,О, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a OH o b ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
As CEO of Aqua Marine, Carrie Easton knows it is important to control costs and to respond quickly to changes in the highly competitive boat-building industry. When Rose Consulting proposes that Aqua...
-
A level setup midway between X and Y reads 6.29 ft on X and 7.91 ft on Y. When moved within a few feet of X, readings of 5.18 ft on X and 6.76 ft on Y are recorded. What is the true elevation...
-
What mix of inputs should be used to produce a given level of output?
-
Joanna takes a security interest in the equipment in Jason Store and files a financing statement claiming equipment and all after acquired equipment. Berkeley later sells Jason Store a cash register...
-
Berset Inc. (BI) has just issued semi-annual coupon bonds with 12 years to maturity and a face value of $1,000 per bond. The bonds make semi-annual coupon payments of $60. What is the price of one...
-
Use Microsoft Visio or Visible Analyst to view and print the contextlevel data flow diagram for the computer inventory system as Chip and Anna did E2 Use the Repository feature or the Repository web...
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
A student combines 60.0 mL of 0.250 M NaOH with 60.0 mL of 0.125 M NaOH. What is the NaOH molar concentration in the resulting solution?
-
3. Create a spreadsheet modeling trajectories of geometric Brownian motion starting at 100 with a growth rate of 5 percent, which is also a risk-free rate, and volatility of 30 percent. Make a...
-
Describe how ethics can play a role in how messages are communicated as well as how they are perceived. Locate a real-world example not found in this module's resources and provide a brief summary of...
-
What is empowerment? How would you define empowerment? Are there disadvantages to empowering employees? What are the benefits of empowering employees?
-
Why is Mixed-Methods Research appropriate for Sears retailing company? Why other methods would not be appropriate?
-
Determine the acceptability of the project using payback period. Year Project "Y" 10 (25,000) 1st year 1,500 2nd Year 2,500 3rd year 5,000 4th year 10,000 5th Year 12,000
-
This chapter introduces you to the many constitutional principles that govern business activities. One such principle is free speech and the extent to which it applies to commercial speech. For...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
Calculate the mass of sodium acetate that must be added to 500.0 mL of 0.200 M acetic acid to form a pH = 5.00 buffer solution.
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
Account for the fact that p-nitro aniline (pKa = 1.0) is less basic than m-nitro aniline (pKa = 2.5) by a factor of 30. Draw resonance structures to support your argument. (The PKa values refer to...
-
What are the main ethical issues that researchers may encounter when conducting business research and how can they address these issues ?
-
How has womens participation in the Olympics changed over the years? Explain
-
What is the Christian view of work according to the God is Not One: The Eight Rival Religions That Run the World.

Study smarter with the SolutionInn App


