Show the products of thesereactions: Brs , b) H,SO, HCI . d) PhC=CPh H,SO4 H9SO, CH,
Question:
Show the products of thesereactions:
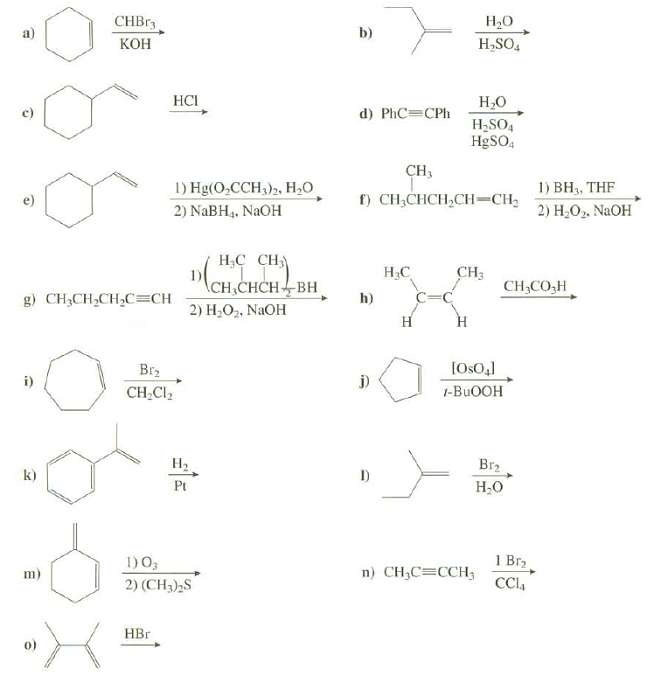
Transcribed Image Text:
СНBrs Н,о b) КОН H,SO, HCI Н.о d) PhC=CPh H,SO4 H9SO, CH, 1) Hg(O,CCH,)2, H,0 1) BH, THF f) CH;CHCH,CH=CH, e) 2) Н.О» NaOH 2) NABH4, NaOH Н.С Сн) CH; CH,CO,H CH CHCHBH h) g) CH,CH,CH,C=CH 2) H,O2, NAOH н Н [Os0,] Br2 j) i) CH.Cl2 1-BUOOH Н. Br2 k) I) Pt H;0 1 Br2 1)0; n) CH,C=CCH; m) CCI 2) (CH3),S HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
All chiral products are produced as ...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Use the DerivaGem software to value a five-year collar that guarantees that the maximum and minimum interest rates on a LIBOR-based loan (with quarterly resets) are 7% and 5% respectively. The LIBOR...
-
What is social media analytics? What are the reasons behind its increasing popularity?
-
The hollow, spherical glass shell shown in FIGURE 15-52 has an inner radius R and an outer radius 1.2R. The density of the glass is (g. What fraction of the shell is submerged when it floats in a...
-
A perpetual option is one that never expires. (Such an option must be of American style.) Use Exercise 6 to show that the value of a perpetual call on a non-dividend-paying stock is \(C=S\). Data...
-
On December 31, 2006 Marsh Company held 1,000 shares of X Company common stock in its portfolio of long-term investments in available-for-sale securities. The stock had cost $15 per share and has a...
-
Mitre Company acquired Midwest Transportation Company for $1,399,100. The fair market values of the assets acquired were as follows. No liabilities were assumed. Equipment Land Building Franchise...
-
The diagram opposite gives details about a companys 115 employees. For example, it employs four unqualified, part-time females. Two employees are selected at random. Find the probability that: a. One...
-
Show the products of these reactions: CH;CH2 CH3 a) b) Pt . Cl2 . d) H,SO4 CCl . . e) CH,CH,CH,C=CH f) H,SO, HgSO, H,SO, CH3 Br2 g) , CH3 h) CH CH3 CI CH3 . 1) BH3, THF 2) H2O2, NAOH CH,CH3 ...
-
Show the products of these reactions of 1-propylcyclopentene with thesereagents: a) Br, CCl, b) Br2, H2O d) HBr f) [OsO,], t-BuOOH c) 1) BH3, THF; 2) H,O,, NaOH e) H2O, H,SO, g) 1) O; 2) (CHz)S
-
A 12.5-L scuba diving tank contains a heliumoxygen (heliox) mixture made up of 24.2 g of He and 4.32 g of O 2 at 298 K. Calculate the mole fraction and partial pressure of each component in the...
-
conduct Quantitative Analysis. you will use Excel and the IBM Cognos tool to analyze and display data to solve problems and inform decision making. Using a method similar to the qualitative research...
-
One powerful but rarely used source of business ideas are: family and friends. work experiences. existing customers. government agencies.
-
Cleaning of equipment is one of the duties for Sally and Elaine. Why is surface cleaning of equipment important?
-
26. In the following scenario, complete the chart by calculating the employee's Canada Pension Plan contribution the employer's Canada Pension Plan contribution the employee's Employment Insurance...
-
You assume that you form a team. Your team is going to take part in an HKMU competition. The maximum number of students in a team is 2. In this competition, your team is responsible for finding a...
-
Some economists believe that during a recession, business demand for investment in factories, office buildings, and machinery becomes less sensitive to changes in the real interest rate. If these...
-
Vince, Inc. has developed and patented a new laser disc reading device that will be marketed internationally. Which of the following factors should Vince consider in pricing the device? I. Quality of...
-
The K b values for ammonia and methylamine are 1.8 10 -5 and 4.4 10 -4 , respectively. Which is the stronger acid, NH 4 + or CH 3 NH 3 + ?
-
Show the product you would obtain from the reaction of cellobiose with the following reagents: (a) NaBH4 (b) Br2 H2O (c) CH3COCl, pyridine
-
Identify the following aldoses, and tell whether each is a D or Lsugar: (a) (b)
-
Draw Fischer projections of the following molecules, placing the carbonyl group at the top in the usual way. Identify each as a D or Lsugar. (b) la)
-
4. Methane burns in air with an equivalence ratio of =0.85, determine the composition of the products: What are the Mass Fractions of each species in the Products?
-
1. Describe the health and safety requirements of the area in which the installation activity is to take place, and the responsibility these requirements place on the learner 2. Describe the...
-
1. Which is easier for nucleation in solids, homogeneous or heterogeneous nucleation? What are the common defects present in solid solution (list at least five kinds of defects)? Are the defects...

Study smarter with the SolutionInn App


