Some of the most important metals we use are found in the transition element section of the
Question:
Some of the most important metals we use are found in the transition element section of the Periodic Table. One of these elements is copper. Sodium, a Group I metal, has very different properties from those of copper. Complete the table below to show their differences.
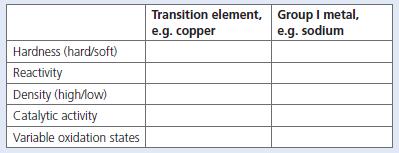
Transition element, Group I metal, e.g. sodium e.g. copper Hardness (hard/soft) Reactivity Density (high/low) Catalytic activity Variable oxidation states
Step by Step Answer:
Transition element copper soft malleable and ductile Copper is low on reactivi...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Some of the most important organic compounds in biochemistry are the _-amino acids, represented by the general formula shown.
-
The following are some of the most important trigonometric identities. Check them at a. = 0, b. = /4, c. = /2, d. = . cos (/2) = 1 + cos()/2. Only check at points a, c, and d.
-
The following are some of the most important trigonometric identities. Check them at a. = 0, b. = /4, c. = /2, d. = . sin2() + cos2() = l.
-
Suppose treacle is an array of 10 floats. Declare a pointer that points to the first element of treacle and use the pointer to display the first and last elements of the array.
-
For each of the following, write the complete chemical equation for the acidbase reaction that occurs. Describe each using Brnsted language (if appropriate) and then using Lewis language (show...
-
Some studies have shown that in the United States, men spend more than women buying gifts and cards on Valentine's Day. Suppose a researcher wants to test this hypothesis by randomly sampling nine...
-
A particle that has mass \(m\) and charge \(q\) enters a uniform magnetic field that has magnitude \(B\) and is directed along the \(x\) axis. The initial velocity of the particle is in the \(x y\)...
-
a. Draw a rune line depicting all of the cash flows associated with Sunrises view of the retirement annuity. b. How large a sum must Sunrise accumulate by the end of year 12 to provide the 20-year,...
-
8.The following data is available: Country Y currency Dollar Country X currency Peso Country Y interest rate 1% per year Country X interest rate 3% per year Country X expected inflation rate 2% per...
-
The bending moment acting on the W360 x 262 section is 460 kN m. Find the maximum bending stress in (a) the flanges; and (b) the web. See Appendix B-2 for properties of the section. Flange W 360 x...
-
Use the following list of metals to answer the questions a to i: iron, calcium, potassium, gold, aluminium, magnesium, sodium, zinc, platinum, titanium. a. Which of the metals is found native? b....
-
a. Consider the chemical properties and physical properties of the halogens chlorine, bromine and iodine. Using these properties, predict the following about the other two halogens, fluorine and...
-
In Exercises the graph of a velocity function is shown. It represents the velocity in miles per hour during a 10-minute trip to work. Make a sketch of the corresponding position function. Velocity...
-
You deposit $100 each month into an account earning 4% interest compounded monthly. Round to nearest cent as needed. a) How much will you have in the account in 15 years? $ b) How much total money...
-
You have just become the manager of an insurance office with five professional agents and several clerical assistants. The office is part of a larger company headquartered in another city. Your...
-
Two cities, New York and Chicago are experiencing a tremendous upsurge in criminal activity ever since the defund the police campaign started. There is competition between the two cities to decrease...
-
A distributor of electrical switches is interested in implementing a total quality program. Following data is collected about the organizations receiving process. Date Packing Slips Number of Errors...
-
Given the company's Vision, Mission, and Objectives (VMO), identify the company's core competencies and assess which ones are rare, costly, or not easily imitated. Discuss how they are related to and...
-
Show that the positive quadrant Q = {(x,y) | x, y > 0) R2 forms a vector space if we define addition by (x1, y1) + (x2, > y2) = (x1 x2, y1 y2) and scalar multiplication by c (x, y) = (xc, yc).
-
Doorharmony Company makes doorbells. It has a weighted- average cost of capital of 5% and total assets of $ 5,900,000. Doorharmony has current liabilities of $ 750,000. Its operating income for the...
-
When 2.25 mg of anthracene, CI4HlO(s), was burned in a bomb calorimeter the temperature raised by 1.35 K. Calculate the calorimeter constant. By how much will the temperature rise when 135 mg of...
-
Calculate the standard enthalpy of solution of AgBr(s) in water from the enthalpies of formation of the solid and the aqueous ions.
-
Given that the standard enthalpy of combustion of graphite is -393.51 kJ mol-1 and that of diamond is -395.41 kJ mol-1, calculate the enthalpy of the graphite-to-diamond transition.
-
Which type of decision making approach believes that those who are affected by a decision should be invited to participate in the decision making?
-
Write this note professionally: To whom it may concern (Human Resources). My name is Clara Shamatanga currently RN 2 former RN3 (Nurse Educator). I recently applied for a Assistant Director of...
-
What is planning and explain it in-depth? What are the basic elements of planning? provide Example

Study smarter with the SolutionInn App


