The lattice energy of magnesium bromide, MgBr 2 , can be calculated using the enthalpy changes shown
Question:
The lattice energy of magnesium bromide, MgBr2, can be calculated using the enthalpy changes shown in the table.
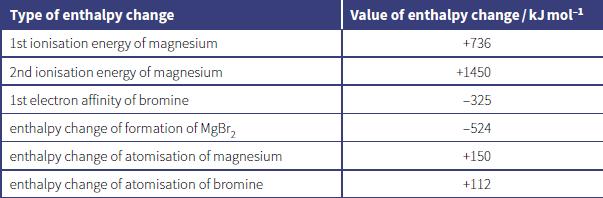
a. State the meaning of the terms:
i. Lattice energy
ii. 2nd ionisation energy.
b. Draw and label a Born–Haber cycle to calculate the lattice energy of magnesium bromide.
c. Calculate the lattice energy of magnesium bromide.
Transcribed Image Text:
Type of enthalpy change Value of enthalpy change / kJ molH 1st ionisation energy of magnesium +736 2nd ionisation energy of magnesium +1450 1st electron affinity of bromine -325 enthalpy change of formation of MgBr, -524 enthalpy change of atomisation of magnesium +150 enthalpy change of atomisation of bromine +112
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a iEnergy change when one mole of an ionic compound forms fr...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
A stock's beta coefficient can be calculated using the following equation: B- = ^2 Vi, a. Write a user-defined function that can calculate the beta coefficient. The arguments to the function should...
-
The lattice energy of an ionic solid such as NaCl is the enthalpy change H° for the process in which the solid changes to ions. For example, NaCl(s) Na+(g) + Cl(g) H = 786 kJ/mol Assume that the...
-
The lattice energy of sodium chloride can be calculated using the following enthalpy changes: Enthalpy change of formation of sodium chloride Enthalpy changes of atomisation of sodium and chlorine ...
-
SG Company acquired 80% of Popsters Company on January 1, 2019, when the stockholders equity of Popsters consisted of: Ordinary shares, P100 par P500,000 Paid in capital in excess of par 400,000...
-
What is the meaning of the term statistical inference? What
-
Derive the binomial distribution in the following algebraic way, which does not involve any explicit combinatorial analysis. One is again interested in finding the probability W(n) of n successes out...
-
In Example 6.3, what is the change in the cart's kinetic energy due to the shove (a) in the Earth reference frame, (b) in a reference frame moving in the same direction as the cart at \(0.60...
-
On January 1, 2014, Christel Madan Corporation had inventory of $50,000. At December 31, 2014, Christel Madan had the following account balances. Freight-in ................ $ 4,000 Purchases...
-
(a) An investor purchased 300 units of a Mutual Fund at Rs. 12.25 per unit on 31 December, 2009. As on 31st December, 2010 he has received Rs. 1.25 as dividend and Rs. 1.00 as capital gains...
-
On August 1, year 1, Hampton Construction received a 9 percent, 6-month note receivable from Dusty Roads, one of Hampton Construction's problem credit customers. Roads had owed $43,200 on an...
-
a. Write equations to represent: i. The 1st ionisation energy of caesium ii. The 3rd ionisation energy of aluminium iii. The enthalpy change of formation of calcium oxide iv. The enthalpy change of...
-
a. Draw a fully labelled BornHaber cycle for potassium bromide, naming each step. b. State the name of the enthalpy changes represented by the following equations: i. I 2 (s) I(g) ii. N(g) + e N ...
-
Consider the following regression equation: a. Predict the value of Y when X1 = 8.5 and X2 = 5.2. b. Interpret the meaning of the regression coefficients b0, b1, and b2. log , = log 3.07 + 0.9 log Xy...
-
Dose healthcare organizations have educational videos to help employees make insurance decisions? Who helps them wade through all of the information out there? How do we make sure that the people who...
-
How does Green Credit act as a creditor and as a debtor in this scenario? Who would be Green Credit's creditor(s) and debtor(s)?
-
Johnson Department Store had net sales of $23 million and cost of goods sold of $12 million for the year. The beginning inventory for the year was $6 million. The ending inventory for the year was $1...
-
Online Vegan Sushi catering business - Evaluate your company's strengths, weaknesses, opportunities, and threats by conducting a SWOT Analysis. - Determine the main sources and intensity of...
-
What is the amount of time it takes for the investment to pay for itself from the financial benefits?
-
A fly with a mass of 1.0 10 4 kg crawls across a table at a speed of 2 mm/s. Compute the de Broglie wavelength of the fly and compare it with the size of a proton (about 1 fm, 1 fm = 1015 m).
-
a) Show that (a, b) := {{a}, {b}} does not satisfy the ordered pair axiom. b) Determine whether each of the following statements is true or false. (Give a reason in each case): (i) {a, b} C (a, b)....
-
The initiation step for radical addition of HBr is highly endothermic: (a) Explain how this step can be thermodynamically favorable at high temperature even though it is endothermic. (b) Explain why...
-
Between 0°C and 100°C, the heat capacity of Hg(l) is given by Calculate ÎH and ÎS if 2.25 moles of Hg(l) is raised in temperature from 0.00° to 88.0°C at constant P....
-
Draw all resonance structures for each of the following radicals: (a) (b) (c) (d) (e)
-
Solve the following LPP graphically, Max Z5x, + 3x Subject to, 3x + 5x2 15 5x, + 2x2 10 XX 0
-
The population standard deviation for the temperature of beers found in Scooter's Tavern is 0 . 3 6 degrees. If we want to be 9 9 % confident that the sample mean beer temperature is within 0 . 1...
-
Probability Scores 0.18 1 0.12 3 0.14 4 0.12 7 0.18 8 0.26 10 Find the expected value of the above random variable.

Study smarter with the SolutionInn App


