Redo Problem 13.10 using Aspen Plus. Problem 13.10 Hydrogen gas can be produced by the following reactions
Question:
Redo Problem 13.10 using Aspen Plus.
Problem 13.10
Hydrogen gas can be produced by the following reactions between propane and steam in the presence of a nickel catalyst:
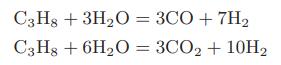
a. Compute the standard heat of reaction and the standard-state Gibbs energy change on reaction for each of the reactions at 1000 and 1100 K.
b. What is the equilibrium composition of the product gas at 1000 K and 1 bar if the inlet to the catalytic reactor is pure propane and steam in a 1-to-10 ratio?
c. Repeat calculation (b) at 1100 K
Transcribed Image Text:
C3H8 + 3H₂O = 3CO + 7H₂ C3H8 + 6H₂O = 3CO₂ + 10H₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
here is the solution to Problem 1310 using Aspen Plus a Standard heat of reaction and standardstate Gibbs energy change The standard heat of reaction ...View the full answer

Answered By

Nicole omwa
Being a highly skilled tutor with at least 5 years of tutoring experience in different areas, I learned how to help diverse learners in writing drafts of research papers, actual research papers and locate credible sources. My assurance is built upon my varied knowledge of a variety of subjects. Furthermore, my involvement and interaction with numerous learners of all levels has allowed me to understand my clients' specific demands. Ultimately, this has aided me in being a better coach to learners to better their grades. Essentially, my responsibilities as a tutor would include:
Teaching abilities that assist pupils in enhancing their academic performance
Personal interaction with learners to make them understand abstract concepts
Inducing new skills and knowledge into their academic journeys
Fostering individual reflection, and independent and critical thinking
Editing and proofreading
Because I am constantly available to respond to your queries, you may decide to rely on me whenever you require my assistance. As an assurance, my knowledge skills and expertise enable me to quickly assist learners with different academic challenges in areas with difficulty in understanding. Ultimately, I believe that I am a reliable tutor concerned about my learner's needs and interests to solve their urgent projects. My purpose is always to assist them in comprehending abstract schoolwork and mastering their subjects. I also understand that plagiarism is a severe offense and has serious ramifications. Owing to this, I always make it a point to educate learners on the numerous strategies to have uniquely unique solutions. I am familiar with the following formatting styles:
MLA
APA
Harvard
Chicago
IEEE
Communication is always the key in every interaction with my learners. Hence, I provide timely communication about the progress of assigned projects. As a result, I make sure that I maintain excellent communication with all of my clients. I can engage with all of my customers more effectively, assisting them with their unique academic demands. Furthermore, I attempt to establish a solid working relationship with my leaners I have exceptional abilities in the below areas;
Sociology
History
Nursing
Psychology
Literature
Health and Medicine
Chemistry
Biology
Management
Marketing
Business
Earth Science
Environmental Studies
Education
Being a teacher who aces in diverse fields, I provide various academic tasks, which include;
Academic Reports
Movie Reviews
Literature Reviews
Annotated bibliographies
Lab reports
Discussion posts
Dissertations
Case study analyses
Research proposals
Argumentative Essays
I guarantee you high-quality Papers!!!!!
5.00+
17+ Reviews
32+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Write and balance the chemical reaction of carbon monoxide forming solid carbon and carbon dioxide vapor. Determine the equilibrium constant at 700 K and 750 K. Will solid carbon form at the...
-
Hydrogen gas can be produced by the following reactions between propane and steam in the presence of a nickel catalyst: Neglecting any other competing reactions: (a) Compute the equilibrium constants...
-
Synthesis gas can be produced by the catalytic re-forming of methane with steam. The reactions are: CH 4 (g) + H 2 O(g) CO(g) + 3H 2 g) CO(g) + H 2 O(g) CO 2 (g) + H 2 (g) Assume equilibrium is...
-
Find an equation in cylindrical coordinates for the equation given in rectangular coordinates. y = x
-
Friendly Freddies is an independently owned major appliance and electronics discount chain with seven stores in a Midwest metropolitan area. Rapid expansion has created the need for careful planning...
-
The following balances have been extracted from the books of the David and Peter Manufacturing Company as at 30 April 2011: 000 Direct wages ............... 70 Factory equipment: at cost...
-
In Exercises 1518, use the given set of points to a. Compute b1. b. Compute the residual standard deviation se. c. Compute the sum of squares for x, (x x)2. d. Compute the standard error of b1, sb....
-
Flores Chemical Co. recorded costs for the month of $18,900 for materials, $44,100 for labor, and $26,250 for factory overhead. There was no beginning work in process, 8,000 units were finished, and...
-
Examine the principles of advanced soil stabilization techniques, including chemical grouting, deep soil mixing, and electro kinetic stabilization, and their applications in mitigating slope...
-
Redo Problem 13.5 using Aspen Plus. Problem 13.5 The production of NO by the direct oxidation of nitrogen, occurs naturally in internal combustion engines. This reaction is also used to commercially...
-
Redo Problem 13.11 using Aspen Plus. Problem 13.11 An important step in the manufacture of sulfuric acid is the gas-phase oxidation reaction Compute the equilibrium conversion of sulfur dioxide to...
-
A utility is submitting their petition to their regulatory agency to justify rates for the upcoming year. Their proposal is based upon revenue requirements. The company has 45 percent of their...
-
Leveraged Investing with Eligible Dividends $50,000 Cash purchase + $50,000 borrowed funds at 4.95% 12 month holding period Eligible monthly dividend of $0.07/share Purchase price of $25.00 Sale...
-
What will be a great strategic recommendation to overcome Buyer Power for T-Mobile. The strategic recommendation needs to be something that has not been proposed before. Any suggestions will help.
-
(b) (i) (ii) (iii) (iv) A symmetrical double-wedge aerofoil has a chord of 50 cm and a thickness of 10%. It is tested in a wind tunnel at Mach 2.9 at an angle of incidence of 3.5 degrees. Use...
-
2. Compute 1/1 + 1/2 + 1/3 + ... + 1/999 + 1/1000. 3. Compute 1 + a/1! + a/2! + a/3! + ... + a100/100!. (Note: cumprod is useful here. Do not use the function factorial.) Compare the result with exp...
-
The Playfair cipher with keyword SWITZERLAND and matrix starting at the top center given below 71 7 71 71 71 . Find the message. 71 . K 71 71 K 71 71 were used to create the following ciphertext: ....
-
Around the world, utilities generally have the highest dividend payouts of any industry, yet they also tend to have massive investment programs which they finance using external sources. How do you...
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
If the pressure in the tire on your car is 32.0 lbf/in 2 (or psi), what is its pressure in SI units? Assume that on the surface of the Earth, g = 9.81 m/s 2 = 32.2 ft/s 2 (i.e., each to three...
-
Suppose the mass in Example 2.1 was 50.0 slugs. What would be its weight in lbf (pounds force)? Assume that on the surface of the Earth, g = 9.81 m/s 2 = 32.2 ft/s 2 (i.e., each to three significant...
-
What would the 5.00 slug mass in Example 2.1 weigh on the Moon where the acceleration of gravity is only 1/6 of that on Earth? Assume that on the surface of the Earth, g = 9.81 m/s 2 = 32.2 ft/s 2...
-
Define the term 'personal development' using your own words and identify at least five personal development skills. Identify at least five professional development activities. Identify and briefly...
-
Please Read two of this pdf and find me 5 highlight differences between their 2 companies in their Code of Business Conduct and Ethics: 1.Madrigal...
-
1. How do company rules, company culture and behaviour guidelines influence how you communicate and manage your team in the workplace? Give an example of a policy and how it works. Answer: Question 2...

Study smarter with the SolutionInn App


