One step in the preparation of nicotine sulfate from tobacco is the extraction of nicotine from aqueous
Question:
One step in the preparation of nicotine sulfate from tobacco is the extraction of nicotine from aqueous solution by kerosene. Table 10.P1 shows equilibrium data for the distribution of nicotine between water and kerosene. Compute the kerosene water ratio required to remove 90 percent of the nicotine from the aqueous stream in a single equilibrium stage.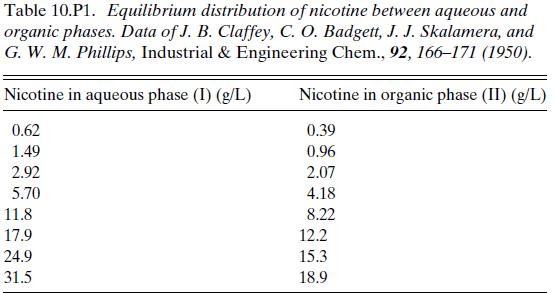
Transcribed Image Text:
Table 10.P1. Equilibrium distribution of nicotine between aqueous and organic phases. Data of J. B. Claffey, C. O. Badgett, J. J. Skalamera, and G. W. M. Phillips, Industrial & Engineering Chem., 92, 166-171 (1950). Nicotine in aqueous phase (I) (g/L) 0.62 1.49 2.92 5.70 11.8 17.9 24.9 31.5 Nicotine in organic phase (II) (g/L) 0.39 0.96 2.07 4.18 8.22 12.2 15.3 18.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the kerosenewater ratio required to remove 90 percent of the nicotine from the aqueous stream in a single equilibrium stage we can use th...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
135+ Reviews
427+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Trace amounts of phenol are to be extracted from an aqueous stream (Phase I) using pure xylene as a solvent. From data in the Chemical Engineers Handbook we have M = 1.4. a. What xylene/water flow...
-
The Business School at Eastern College is collecting data as a first step in the preparation of next years budget. One cost that is being looked at closely is administrative staff as a function of...
-
The first step in the preparation of lead from its ore (galena, PbS) consists of roasting the ore. 2PbS(s) + 3O2(g) 2SO2(g) + 2PbO(s) Calculate the standard enthalpy change for this reaction, using...
-
(a) Compute the primary Class 1 NICs payable by the following weekly-paid employees for the week ending 14 August 2020: (i) Employee A has earnings for the week of 110. (ii) Employee B has earnings...
-
The following are various activities an auditor does during audit planning. 1. Send an engagement letter to the client. 2. Tour the client's plant and offices. 3. Compare key ratios for the company...
-
An article in the Wall Street Journal discussed why the hotel workers' union in New York City was against a proposal for more hotels to be built in Midtown Manhattan: "The union is concerned that...
-
Data set: Systolic blood pressure levels (in millimeters of mercury) of 28 patients Construct a frequency distribution and a relative frequency histogram for the data set using five classes. Which...
-
Go to www.petrobras.com, click on About us, and read about the oil company. What are the key pressures for change reported in these statements?
-
Margo, a calendar year taxpayer, paid $1,580,000 for new machinery (seven-year recovery property) placed in service on August 1, 2023. Use Table 7-2. Required: a. Assuming that the machinery was the...
-
The data in Table 10.P2 are from a batch experiment measuring the distribution of octanoic acid between an aqueous phase consisting of a solution of corn syrup in water and an organic xylene phase....
-
In analogy to the treatment in Section 5.7, derive the equations describing a countercurrent membrane reactor in which the reversible first-order reaction A M is carried out. Section 5.7: The staged...
-
Sylvia Chan is a clerk in the shoe department of the Hudson's Bay store in Winnipeg. She earns a base monthly salary of $1,875 plus a 7 percent commission on her sales. Through payroll deductions,...
-
How does lean production impact the overall safety and risk management practices within an organization?
-
Water, initially saturated vapor at 11.5 bar, fills a closed, rigid container. The water is heated until its temperature is 200C. For the water, determine the pressure at the final state, in kPa.
-
How do bureaucratic processes and procedures impact the day to day operations of school?
-
The v s graph for a test vehicle is shown. Determine its acceleration and the time required for the vehicle to travel a distance of s= 100 m and s = 175 m if s = 0 when t = 0. v (m/s) 50 150 200 s (m)
-
Students can feel more targeted if the single act is highlighted. I believe having a system that targets bullying as a whole can bring the community together to follow the system as a unit. By using...
-
What is meant by the term rubber stamp supervisor and how are these individuals utilized in a payroll fraud scheme?
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
A component A spreads from a point source as a result of Brownian motion in one dimension. At time t = 0, all molecules are located at x = 0. During a 0.5 s time step, each molecule has the same...
-
Ethylene is dimerized to 1-butene on the surface of a catalyst. At high temperature the reaction kinetics becomes very fast and the rate of reaction becomes controlled by diffusional mass transfer....
-
Under certain conditions, evaporation of a solvent A from freshly-applied paint into air is controlled by diffusion. Assume that diffusion occurs through a thin film of stagnant air adjacent to the...
-
Car A and car B travel in the same direction along a straight section of the interstate highway. For the entire interval shown on the velocity-versus-time graph (see figure below), car A is ahead of...
-
A novice golfer on the green takes three strokes to sink the ball. The successive displacements of the ball are d = 4.02 m to the north, d = 2.10 m northeast, and d3 = 1.18 m at 30.0 west of south
-
A point particle with charge q = = -200 nC and mass m = 3.0 106 g is released from rest at the midpoint of a parallel-plate capacitor. The capacitor has plate separation d 3.0 mm. The negative...

Study smarter with the SolutionInn App


