Calculate the equilibrium constants for the following reactions: 2+ (a) 2 Fe+ (aq) + H(g) 2 Fe+
Question:
Calculate the equilibrium constants for the following reactions: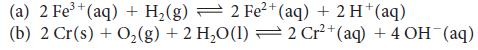
Transcribed Image Text:
2+ (a) 2 Fe³+ (aq) + H₂(g) 2 Fe²+ (aq) + 2 H+(aq) (b) 2 Cr(s) + O₂(g) + 2 H₂O(l) — 2 Cr²+ (aq) + 4 OH (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To calculate the equilibrium constants K for the given reactions we use the ge...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Calculate the equilibrium constants of the following reactions at 25C from standard potential data: (a) Sn(s) + CuS04 (aq) ~ Cu(s) + SnS04 (aq) (b) Cu2+(aq) + Cu(s) ~ 2 Cu+{aq)
-
Predict whether the equilibrium constants for the following reactions should be greater than 1 or less than 1: (a) CdI(s) + CaF(s) CdF(s) + Cal,(s) (b) [Cul 1-(aq) + [CuCl](aq) (c) NH;(aq)...
-
If two reactions sum to an overall reaction, and the equilibrium constants for the two reactions are K 1 and K 2 , what is the equilibrium constant for the overall reaction?
-
Suppose you make beautiful coffee tables out of juniper trees. Your materials cost for each table is $135. You pay your craftsman $25 per hour and it takes him 5 hours to complete one table. If you...
-
Opportunity costs. The Wolverine Corporation is working at full production capacity producing 10,000 units of a unique product Rosebo. Manufacturing cost per unit for Rosebo is as follows:...
-
South Carolina Furniture, Inc., produces all types of office furniture. The Executive Secretary is a chair that has been designed using ergonomics to provide comfort during long work hours. The chair...
-
Car Sales. Consider again the data on used cars (ToyotaCorolla.jmp) with 1436 records and details on 38 attributes, including Price, Age, KM, HP, and other specifications. The goal is to predict the...
-
Maine Department Store is located near the Village Shopping Mall. At the end of the companys calendar year on December 31, 2010, the following accounts appeared in two of its trial balances....
-
A lottery offers two options for a prize, A and B, as shown below. Option A : $1,000 a week for life. Option B : $1,000,000 in one lump sum. If you choose Option B, you have the opportunity to place...
-
Barium hydroxide is a base that is sometimes used for titrating acids. When using it, you need to know the actual concentration of hydroxide ion in the solution. What are the molar concentrations of...
-
(a) Sketch the titration curve for the titration of 5.00 mL 0.010 m HCl(aq) with 0.010 m Ca(OH) 2 (aq), indicating the pH of the initial and final solutions and the pH at the stoichiometric point....
-
Compute the entropy change of 5.00 g of water at 100 C as it changes to steam at 100 C under standard pressure.
-
Cross regulators in main canals are provided (A) To regulate water supply in the distributaries (B) To increase water head upstream when a main canal is running with low supplies (C) To overflow...
-
Separable PDES Q1.Reduce the following PDE equation to ODES via separable method only(No need to Integrate them) in terms of y=y(x), u=u(x) and u=u(y) (a) (i) , + 4, + 2 0 (ii) 2ur + 5uy5u = 0 (ii)...
-
Q2. Q3. (a) (i) Q4. (a) (i) (i) = (x, y) 8% (a=x0+,y=yo) 0x (x=x0,y=yo) ad = (l,m) ail(l=lo+1,m=mo) ail(l=lo,m=mo) ad * = f(x) 3 d dx2](x=x0+) = {3\x=x0) dx
-
1 Let X ~ Nn(1,), where 1 = and is the variance covariance matrix of X. Let 1 1 0 0 0 1 1 1 1 0 1 0 0 1 1 1 1 = (1 p)I + pJ, with p > -11, I = and J = . Therefore, 0 0 0 1 1 1 1 1 when p = 0 we...
-
Q1.(a) Explain the difference between a signal and a system. Give two areas of application each for a signal and system. (b) Suppose you have been tasked to design an automated system for gas cooker...
-
Worthington Company employs a job-order costing system. Before disposition of the underapplied or overapplied over head, selected year-end balances from Worthingtons accounting records were: Cost of...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Why is it true for any quantum mechanical problem that the set of wave functions is larger than the set of eigenfunctions?
-
By discussing the diffraction of a beam of particles by a single slit, justify the statement that there is no sharp boundary between particle-like and wave-like behavior.
-
Form the operator A 2 if A = d 2 /dy 2 + 3y (d / dy) 5. Be sure to include an arbitrary function on which the operator acts.
-
Using the article provided below, you are required to analyse the company information to define the business goals and evaluate whether the supply chain is a strategic fit with the business and...
-
Create output that shows a scatterplot with a line of best fit for your data. Explain what a line of best fit is? Submit both the output file (SPSS) as well as your Word summary.
-
Activities list and find out the total project duration in total days (You don't need to submit the project diagram, just submit the total days of project duration) What is the Activity Predecessor...

Study smarter with the SolutionInn App


