Calculate the potential of a cell constructed with two nickel electrodes. The electrolyte in one compartment is
Question:
Calculate the potential of a cell constructed with two nickel electrodes. The electrolyte in one compartment is 1.0 m Ni(NO3)2(aq).
In the other compartment, NaOH has been added to a Ni(NO3)2 solution until the pH = 11.0 at 298 K. See Table 6I.1.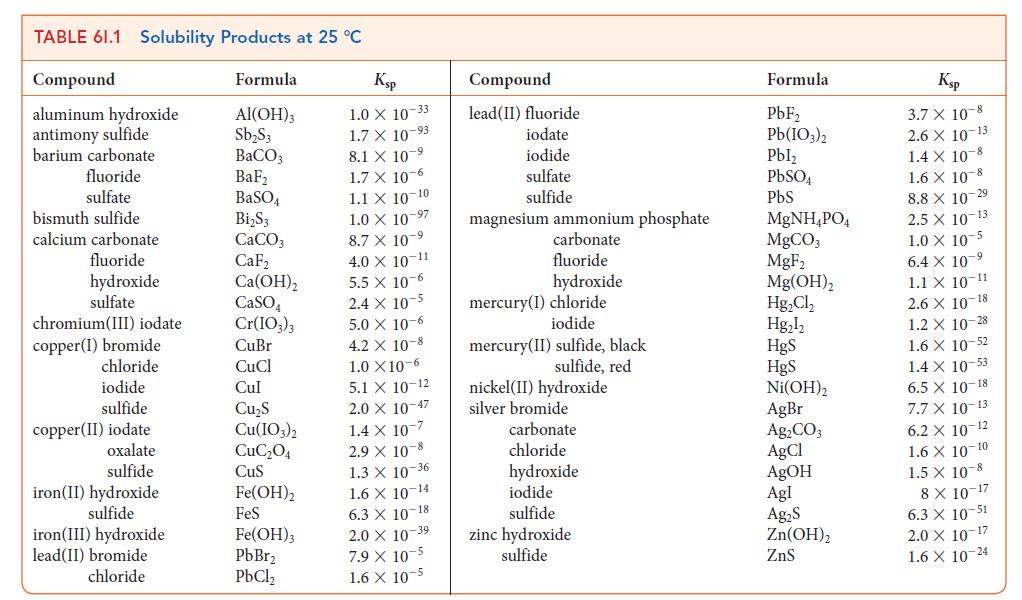
Transcribed Image Text:
TABLE 61.1 Solubility Products at 25 °C Compound aluminum hydroxide antimony sulfide barium carbonate fluoride sulfate bismuth sulfide calcium carbonate fluoride hydroxide sulfate chromium (III) iodate copper (1) bromide chloride iodide sulfide copper(II) iodate oxalate sulfide iron(II) hydroxide sulfide iron(III) hydroxide lead(II) bromide chloride Formula Al(OH)3 Sb₂S3 BaCO3 BaF₂ BaSO4 Bi₂S3 CaCO3 CaF₂ Ca(OH)₂ CaSO4 Cr(IO3)3 CuBr CuCl Cul Cu₂S Cu(IO3)2 CuC₂04 CuS Fe(OH)2 FeS Fe(OH)3 PbBr₂ PbCl₂ Ksp 1.0 X 10-33 1.7 X 10-93 8.1 X 107 1.7 x 10-6 1.1 X 10-10 1.0 X 10-97 8.7 X 10-9 4.0 X 10-11 5.5 x 10-6 2.4 x 10-5 5.0 x 10-6 4.2 X 10-8 1.0 X10-6 5.1 X 10-12 2.0 X 10-47 1.4 x 10-7 2.9 X 10-8 1.3 X 10-36 1.6 X 10-14 6.3 X 10-18 2.0 X 10 39 7.9 X 10-5 1.6 X 10-5 Compound lead(II) fluoride iodate iodide sulfate sulfide magnesium ammonium phosphate carbonate fluoride hydroxide mercury(I) chloride iodide mercury (II) sulfide, black sulfide, red nickel (II) hydroxide silver bromide carbonate chloride hydroxide iodide sulfide zinc hydroxide sulfide Formula PbF₂ Pb(103)2 Pbl₂ PbSO4 PbS MgNH₂PO4 MgCO3 MgF₂ Mg(OH)₂ Hg₂Cl₂ Hg₂l₂ HgS HgS Ni(OH)2 AgBr Ag₂CO3 AgCl AgOH Agl Ag₂S Zn(OH)₂ ZnS Ksp 3.7 X 10-8 2.6 X 10-13 1.4 X 10-8 1.6 X 10 8 8.8 X 10-29 2.5 X 10-13 1.0 X 10-5 6.4 X 10 ⁹ 1.1 X 10-11 2.6 X 10-18 1.2 X 10-28 1.6 X 10-52 1.4 x 10-53 6.5 X 10-18 7.7 X 10-13 6.2 X 10-12 1.6 X 10-10 1.5 X 10-8 8 X 10-17 6.3 X 10-51 2.0 X 10-17 1.6 X 10-24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
A cell was constructed with two lead electrodes. The electrolyte in one compartment is 1.0 m Pb(NO 3 ) 2 (aq). In the other compartment, NaI has been added to a Pb(NO 3 ) 2 solution until a yellow...
-
Calculate the potential of a copper electrode immersed in (a) 0.0380 M Cu(NO3)2. (b) 0.0650 M in NaCl and saturated with CuCl. (c) 0.0350 M in NaOH and saturated with Cu(OH)2. (d) 0.0375 M in...
-
Find the probability | (p)| 2d3 p of the particular momentum p for the ground-state hydrogen atom. (This is a nice exercise in three-dimensional Fourier transforms. To perform the angular...
-
If you are running at top speed toward a source of sound at 1000 Hz, estimate the frequency of the sound that you hear. Suppose that you can recognize a change in frequency of 3%. Can you use your...
-
How does Ajax help to build effective Web pages?
-
A mountain chart is a creative way to display the distribution of a quantitative variable over different categories. The overall distribution is shown as a smoothed histogram, and the area underneath...
-
Eddy's Piano Rebuilding Company has been operating for one year (2010). At the start of 2011, its income statement accounts had zero balances and its balance sheet account balances were as follows:...
-
Blue Industries purchased a machine from Calico Corporation on October 1, 2016. In payment for the $144,000 purchase, Blue issued a one-year installment note to be paid in equal monthly payments at...
-
Data Table 2: Sodium Hypochlorite SDS information Items Physical State Route of Exposure & Symptoms Protective Equipment First Aid Procedures Fire-fighting Measures Chemical Reactivity Safe Storage...
-
The pK a of HIO(aq), hypoiodous acid, is 10.64 and that of HIO 3 (aq), iodic acid, is 0.77. Account for the difference in strength.
-
You find a bottle of a pure silver halide that could be AgCl or AgI. Develop a simple chemical test that would allow you to distinguish which compound was in the bottle.
-
The process of experimenting with base case, best case, and worst case scenarios to see what would happen to company profits under those conditions would be an example of a. Sequencing analysis. b....
-
Two collisions are carried out to crash-test a \(1000-\mathrm{kg}\) car. (a) While moving at \(15 \mathrm{mi} / \mathrm{h}\), the car strikes an identical car initially at rest. (b) While moving at...
-
During much of the postwar period, the Fed attempted to stabilize nominal interest rates. However, during 1979-1982 the Fed under Paul Volcker greatly reduced its emphasis on interest rate...
-
A \(0.20-\mathrm{kg}\) steel ball is dropped into a ball of dough, striking the dough at a speed of \(2.3 \mathrm{~m} / \mathrm{s}\) and coming to rest inside the dough. If it were possible to turn...
-
The positions of two identical carts at rest on a low-friction track are measured relative to two axes oriented in the same direction along the track. On axis \(\mathrm{A}\), cart 1 is at...
-
A flying bug hits the helmet of a coasting bicyclist. (What kind of collision do you expect this to be?) Draw the momentum vectors for the bug and the bicyclist before and after the collision \((a)\)...
-
A random sample of small-business managers was given a leadership style questionnaire. The results were scaled so that each manager received a score for initiative. Suppose the following data are a...
-
Baxter, Inc., owns 90 percent of Wisconsin, Inc., and 20 percent of Cleveland Company. Wisconsin, in turn, holds 60 percent of Clevelands outstanding stock. No excess amortization resulted from these...
-
Several reactions and their standard reaction enthalpies at 298.15 K are given here: The standard enthalpies of combustion of graphite and C 2 H 2 (g) are 393.51 and 1299.58 kJ mol 1 , respectively....
-
For each type of bond below, determine the direction of the expected dipole moment. a) C O b) C Mg c) C N d) C Li e) C Cl f) C H g) O H h) N H
-
Given the following heat capacity data, calculate ÎH o f of CO 2 (g) at 525 K. C(graphite) co:(g) 0:(g) Substance CP.m/J mol-K-1 8.52 28.8 37.1
-
What advancements have been made in the development of multi-objective optimization algorithms that can simultaneously optimize conflicting objectives in fields like environmental conservation and...
-
If AB is parallel to CD where A (2, 3) and B (4, 9), calculate the slope of CD
-
explain the role of surrogate modeling and surrogate-based optimization methods in accelerating the optimization of computationally expensive simulation-based models in fields such as computational...

Study smarter with the SolutionInn App


