Hydrogen cyanide is produced industrially by the following exothermic reaction: Is the high temperature needed for thermodynamic
Question:
Hydrogen cyanide is produced industrially by the following exothermic reaction:
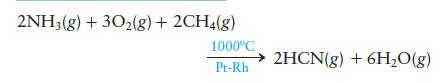
Is the high temperature needed for thermodynamic or for kinetic reasons?
Transcribed Image Text:
2NH3(g) + 302(g) + 2CH4(g) 1000°C Pt-Rh 2HCN(g) + 6H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
The high temperature needed for industrial production of hydrogen cyanide is due to both thermodynamic and kinetic reasons The thermodynamic aspect of ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Hydrogen cyanide is produced industrially by the following exothermic reaction: Is the high temperature needed for thermodynamic or for kinetic reasons? Pr-Rh
-
Hydrogen cyanide is produced industrially from the reaction of gaseous ammonia, oxygen, and methane: 2NH 3 (g) + 3O 2 (g) + 2CH 4 (g) 2HCN( g) + 6H 2 O(g) If 5.00 10 3 kg each of NH 3 , O 2 , and...
-
Ammonia gas is made industrially by the Haber process, which involves the reaction between the gases nitrogen and hydrogen. The amount of ammonia gas produced from this reaction is affected by both...
-
In Exercises 7192, find and simplify the difference quotient f(x +h)-f(x) h -, h = 0
-
You and your roommate are up one night studying microeconomics, and your roommate looks puzzled. You ask what is wrong, and you get this response: The book says that in the short run fixed costs are...
-
In Problem, find the particular solution to each differential equation. y' = 4x 3 + 3x 2 , if y(0) = 4
-
Refer to Samsungs financial statements in Appendix A. Compute its debt ratio as of December 31, 2015, and December 31, 2014. Data From Samsung Financial Statement Appendix A Samsung Electronics Co.,...
-
Best Bathware Company manufactures faucets in a small manufacturing facility. The faucets are made from zinc. Manufacturing has 50 employees. Each employee presently provides 36 hours of labor per...
-
In Milestone Two, you will review the Final Project Client Information document to identify all beneficiaries and discuss potential tax ramifications of receiving inherited property. In addition, you...
-
Banana Computer Company has fixed costs of production of $100,000, while each unit costs $600 of labor and $400 of materials and fuel. At a price of $3000, consumers would buy no Banana computers,...
-
For the reaction 2O(g) O 2 (g) a. Predicts the signs of DH and DS. b. Would the reaction be more spontaneous at high or low temperatures?
-
When most biological enzymes are heated, they lose their catalytic activity. The change Original enzyme new form that occurs upon heating is endothermic and spontaneous. Is the structure of the...
-
A spherical shell of radius R carries a uniform surface charge 0 on the "northern" hemisphere and a uniform surface charge 0 on the "southern" hemisphere. Find the potential inside and outside the...
-
Jennifer and Brad were engaged to be married. They entered into an ante-nuptial agreement pursuant to which they both surrendered any rights to alimony or a property settlement upon divorce. Pursuant...
-
A colorblind man and his wife have four children, 2 boys and 2 girls. One of the boys is colorblind and so is one of the girls. What is the genotype of the mother? What are the genotypes of each of...
-
Write a procedure for Wamco payroll confidentiality and secure storage You are a payroll officer for Wamco. Often family members and other external parties ring you up or email you and want to check...
-
Few athletes were used as endorsers in the 1960s, but that changed dramatically in the 1980s ,name industry began using athletes to endorse their products?
-
A city issues ten-year, $10,000,000 bonds at face value on January 1, 2023, to finance construction of a new school. Calculate the impact of this issuance on the total fund balance in the fund...
-
What are intermediate targets? How do they differ from monetary policy goals? List the two principal types of intermediate targets that the Bank of Canada has used.
-
Write an essay describing the differing approaches of nursing leaders and managers to issues in practice. To complete this assignment, do the following: 1. Select an issue from the following list:...
-
Write electron configurations for the most stable ion formed by each of the elements Rb, Ba, Se, and I (when in stable ionic compounds).
-
Give an example of an ionic compound where both the anion and the cation are isoelectronic with each of the following noble gases. a. Ne b. Ar c. Kr d. Xe
-
What noble gas has the same electron configuration as each of the ions in the following compounds? a. cesium sulfide b. strontium fluoride c. calcium nitride d. aluminum bromide
-
discuss the role of biofilms in microbial growth and persistence, including the molecular mechanisms of biofilm formation, matrix composition, and the implications for antibiotic resistance, host...
-
Monique wants to simplify the expression 3 log 4+3 log 2 She knows that the first step is to write this expression as a single logarithm. If Monique completes the first step correctly what would her...
-
You find PBB Corp's 3.1% bonds at a price quote of ($)97.7 on the finra.org The bond pays semiannually and matures 6 months from now. The bond's YTM is %. You may round your final answer to 4 decimal...

Study smarter with the SolutionInn App


