Name the amino acids in Table 11E.3 that contain nonpolar side groups. These groups contribute to the
Question:
Name the amino acids in Table 11E.3 that contain nonpolar side groups. These groups contribute to the tertiary structure of a protein by preventing contact with water.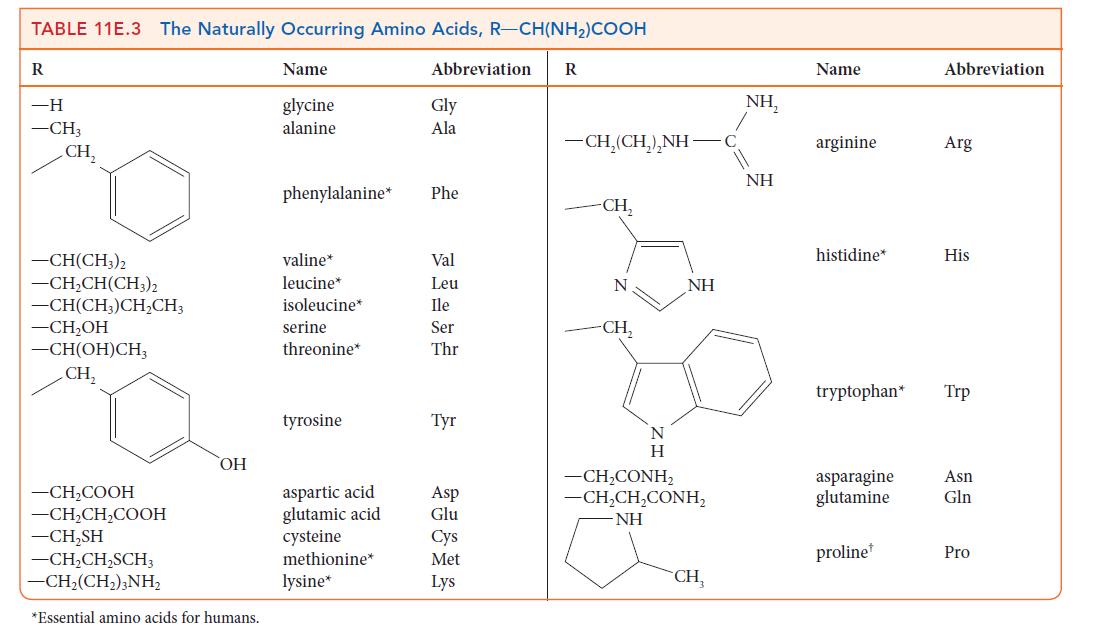
Transcribed Image Text:
TABLE 11E.3 The Naturally Occurring Amino Acids, R-CH(NH)COOH R -H -CH3 CH -CH(CH3)2 -CHCH(CH3)2 -CH(CH3)CHCH3 -CHOH -CH(OH)CH CH, -CHCOOH -CHCHCOOH -CHSH -CHCHSCH3 CH,(CH,),NH, OH *Essential amino acids for humans. Name glycine alanine phenylalanine* valine* leucine* isoleucine* serine threonine* tyrosine aspartic acid glutamic acid cysteine methionine* lysine* Abbreviation Gly Ala Phe Val Leu Ile Ser Thr Tyr Asp Glu Cys Met Lys R CH,(CH,),NH -CH N -CH N H NH CH,CONH, CH,CH,CONH, NH CH NH, NH Name arginine histidine* tryptophan* asparagine glutamine proline Abbreviation Arg His Trp Asn Gln Pro
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
From the image provided Table 11E3 lists the naturally occurring amino acids their structures names ...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Name the amino acids in Table 11E.3 that contain side groups capable of forming hydrogen bonds. This interaction contributes to the tertiary structure of a protein. TABLE 11E.3 The Naturally...
-
Which amino acids in Table 17.1 have nonpolar R groups? Highly polar groups? Relatively flat R groups? Table 17.1 QUESTION CONTINUE TO NEXT PAGE Table 17.1 Names and Formulas of the Common Amino...
-
Let {Wt} be an SBM and define the process {Bt} by Bt = Wt t*W10/10, t [0, 10]. (a) For t [0, 10], what is the probability distribution of Bt? (b) Fix t [0, 10] and consider the bivariate random...
-
Accounting The Case: Patient Khaled is a 75-year-old man admitted to the hospital for a small bowel obstruction. His medical history includes hypertension. Khaled is on NPO. He has a nasogastric (NG)...
-
Use source transformation to determine Io in the network infigure. V2 4/0 A -j1 n 12/0 V 2/00 A
-
Besides comparing a company's performance to its total industry, discuss what other comparisons should be considered within the industry.
-
Discuss the importance of the patients responsibility to communicate to the staff any symptoms he or she is experiencing.
-
Tisdale Incorporated reports the following amount in its December 31, 2015, income statement. Required: 1. Prepare a multiple-step income statement. 2. Explain how analyzing the multiple levels of...
-
Suppose a company had an initial investment of $50,000. The cash flow for the next five years are $18,000, $15,000, $13,000, $16,000, and $17,000, respectively. The interest rate is 11%. What is the...
-
Give the systematic name of each of the following amines: (a) CH 3 NH 2 ; (b) (CH 3 CH 2 ) 2 NH; (c) o-CH 3 C 6 H 4 NH 2 .
-
(a) How many liters of hydrogen at 1.00 atm and 298 K are needed to hydrogenate (i) 1.00 mol C 6 H 10 , cyclohexene; (ii) 1.00 mol C 6 H 6 , benzene, completely? (b) Estimate the reaction enthalpy of...
-
Bitter Sea Ltd. began operations on January 2, 2024. During the year, the following transactions affected shareholders equity: 1. Bitter Seas articles of incorporation authorize the issuance of 2.5...
-
After retaining Jeremiah Jenkins as legal counsel, Bill, indicted for grand larceny, fled the state to save himself from trial and possible conviction. After investigating the facts Bill provided,...
-
La Morenita is a Mexican company dedicated to the production and sale of canned chili peppers. It currently has 30 suppliers, all farmers from the southern region of the state of Tlaxcala; They are...
-
Can users with ther right permissions in a cient - paid subscription add QuickBooks Online Payroll or QuickBooks Time to their QuickBooks Online company?
-
Consider each situation separately. Identify the missing internal control procedure from these characteristics: Assignment of responsibilities Separation of duties Audits Electronic devices Other...
-
a) What is the expected return for Stock A? b) What is the standard deviation for Stock C? c) Which of the three stocks is a rational investor most likely to purchase? d) What is the expected return...
-
Suppose the production possibility frontier for guns (X) and butter (Y) is given by X2 + 2Y2 = 900 a. Graph this frontier. b. If individuals always prefer consumption bundles in which Y = 2X, how...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Each of the following aldehydes was converted into an -amino nitrile followed by hydrolysis to yield an amino acid. In each case, draw and name the amino acid that was produced. (a) Acetaldehyde (b)...
-
Identify the reagents necessary to make each of the following amino acids using a HellVolhardZelinski reaction. (a) Leucine (b) Alanine (c) Valine
-
Draw the aldehyde that is obtained as a byproduct when l-leucine is treated with ninhydrin.
-
Mary Branson invested her summer earnings of $4,000 in a savings account for college. The account pays 3.5% interest. How much will this amount to in 9 months?
-
Brock Company's financial information is as follows. Assume that all balance sheet amounts represent both average and ending balance figures and that all sales were on credit. Assets Cash and...
-
STOCKS There are two investments that have generated the following return in the last four years. Year Return (A) Return (B) 12 15% 30% 2 10% -20% 3 10% -10% 4 5% 40% Both have a return of 10% per...

Study smarter with the SolutionInn App


